Chemistry is the branch of science that revolves around the properties, composition, and structure of elements and compounds, how they change, and the release or absorption of energy.
 Mariyah MatawalaAll the elements and compounds were organized into a framework by the Russian chemist Dmitri Mendeleev in 1869. He organized the elements by atomic weight, where regular and predictable variations in the atomic structure of elements are the cause of periodicity. In the current periodic table, elements in the same group (column) have the same number of valence electrons (outer electrons), and the elements in the same period (row) have the same number of energy levels.
Mariyah MatawalaAll the elements and compounds were organized into a framework by the Russian chemist Dmitri Mendeleev in 1869. He organized the elements by atomic weight, where regular and predictable variations in the atomic structure of elements are the cause of periodicity. In the current periodic table, elements in the same group (column) have the same number of valence electrons (outer electrons), and the elements in the same period (row) have the same number of energy levels.
A key trend in the periodic table is the atomic radius, the distance between the outer shell of an element and the nucleus. From left to right across the period, the atomic radius decreases as the net positive charge experienced by valence electrons increases; the electrons have a stronger attraction to the nucleus, pulling them inwards and closer to it, reducing the distance to the outermost shell. In a group, the atomic radius increases as the number of shield/core electrons and energy levels increases, increasing the distance between the outermost shell and the nucleus (Garrett).
Atomic Radius is highly correlated with Young’s modulus, a key property in determining paint hardness. It is most commonly a measure of the mechanical properties of linear elastic solids, such as rods and wires; it is also known as the elastic modulus or tensile modulus. The value of Young's modulus is most frequently used, but other numbers, such as bulk modulus and shear modulus, are also used to measure a material's elastic properties. Young’s module is most commonly used because it provides details about a material's tensile elasticity, or its capacity to deform along an axis (Britannica, 2023).
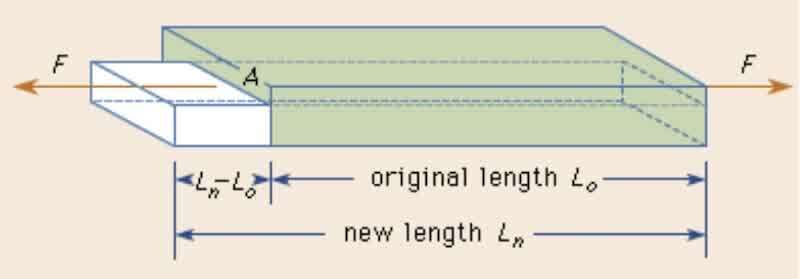 Fig. 1: Explaining Young's modulus (Britannica 2023).
Fig. 1: Explaining Young's modulus (Britannica 2023).
A metal bar with a cross-sectional area of A will stretch from its initial length LI to a new length LN if it is pulled by a force F at each end; the cross-section decreases simultaneously. The stress, expressed as F/A, is the quotient of the cross-sectional area and the tensile force. The strain (has no dimensions) is calculated by change in length, LN - LI, divided by the initial length, (LN - LI)/LI (Britannica, 2023). Hence, Young’s modulus demonstrates the relationship between stress (force per unit area) and strain (proportional deformation in an object). It demonstrates a material's ability to withstand length changes under tension or compression. (Britannica, 2023).
Formula: 𝐸 = s/e
- E is Young’s modulus measured in Pa or N/m2
- sis the stress applied to the material
- eis the strain or deformation corresponding to the stress applied to the material
𝑌𝑜𝑢𝑛𝑔'𝑠 𝑀𝑜𝑑𝑢𝑙𝑢𝑠 = 𝑆𝑡𝑟𝑒𝑠𝑠/Strain = 𝐹𝑜𝑟𝑐𝑒 ÷ 𝐶𝑟𝑜𝑠𝑠 𝑠𝑒𝑐𝑡𝑖𝑜𝑛 𝑎𝑟𝑒𝑎 / [(𝑛𝑒𝑤 𝑙𝑒𝑛𝑔ℎ𝑡 − 𝑖𝑛𝑖𝑡𝑖𝑎𝑙 𝑙𝑒𝑛𝑔ℎ𝑡) ÷ 𝑖𝑛𝑖𝑡𝑖𝑎𝑙 𝑙𝑒𝑛𝑔ℎ𝑡]
When a specific load is placed on a solid object, it deforms. If the object is elastic, the body returns to its initial shape when the pressure is released. Young's modulus only matters in the range where the material reverts to its initial dimensions upon removal of the external force, and the stress is proportionate to the strain. As a large amount of stress is applied to the material, it may flow, experience permanent deformation, or ultimately break (Britannica, 2023). Ergo, after a certain degree of deformation, many materials lose their linear and elastic properties. Young's modulus is constant only for linearly elastic materials (Young’s Modulus, 2020). A material would be more elastic and flexible the larger its value of Y (Aakash Educational Services Limited, 2023). For example, acrylic paint has a lower Young’s modulus, making it more flexible than enamel paint, which exhibits greater stiffness due to its higher modulus.
Young's modulus does not readily apply to liquids or gases; it is primarily defined for solids because the material must have a high density and be in a fixed structure. Only when the material is in a solid state can force be applied to it. For liquids, Young’s modulus can be calculated by using the Bulk modulus (Urone et al., 2012). Paints and protective coatings are engineered materials whose performance depends not only on chemical composition but also on mechanical properties such as stiffness, durability, and resistance to deformation. These properties are influenced by the atomic structure of the inorganic pigments and fillers used within paint films.
One key mechanical parameter governing this behavior is Young’s modulus, as it reflects a material’s resistance to elastic deformation.
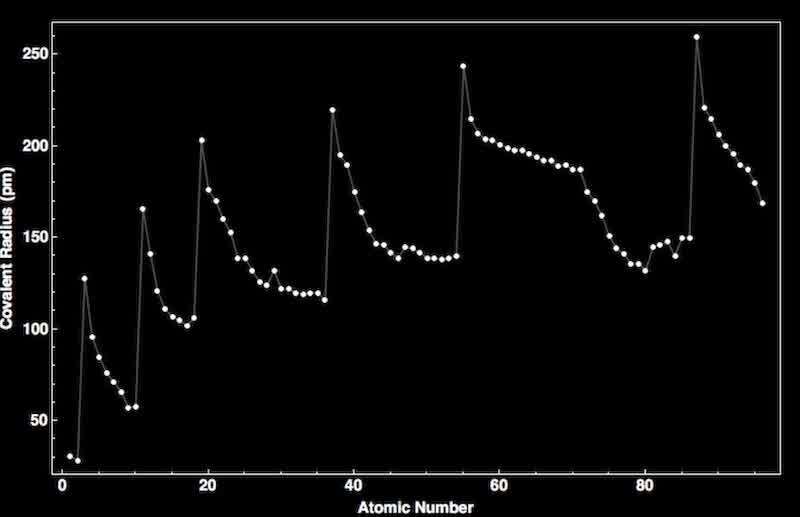
Figure 2: Graph of Atomic Number vs Covalent Radius in picometers (Wolfram Research Inc., n.d.)
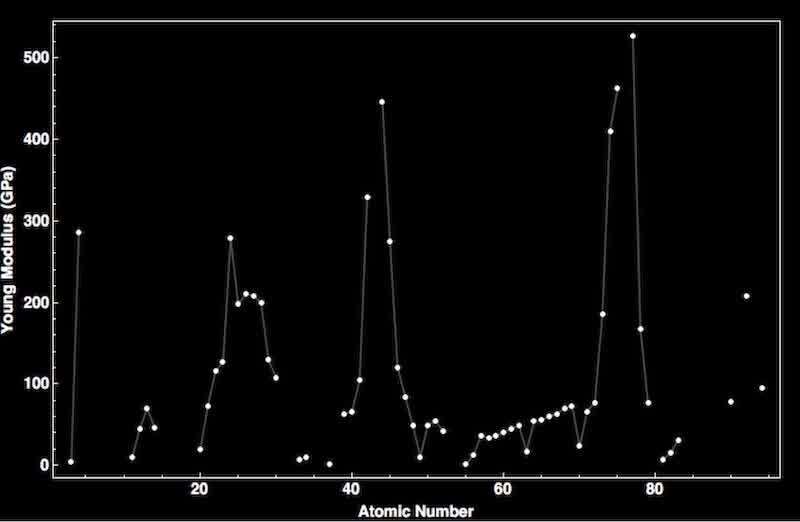
Figure 3: Graph of Atomic Number vs Young’s modulus in GPa (Wolfram Research Inc., n.d.)
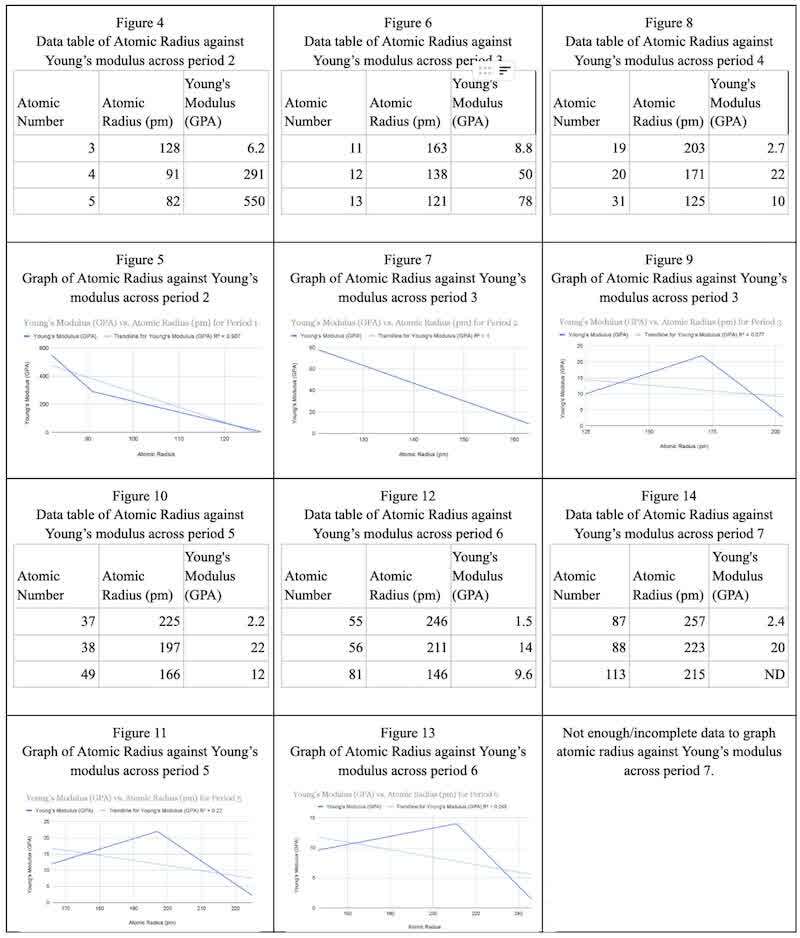
The data above show that the atomic radius and Young’s modulus exhibit a linear negative correlation for periods 2 and 3 (Figures 5 and 7). As the atomic radius increased, Young’s modulus decreased. This negative correlation can be explained by the atomic structure and bonding of the atoms. The elements in period 2 have 2 energy levels/shells, and the elements in period 3 have 3 energy levels/shells. Moving from left to right, the effective nuclear charge increases as the number of protons increases, while the number of energy levels and shielding electrons stays the same. This causes the atomic radius to decrease as the electrons in the outermost energy level are pulled toward the nucleus by its increasing charge. As the atom becomes more compact with a decreasing atomic radius, Young’s modulus decreases because the material can be stretched less and experiences a lower amount of strain. Its elasticity decreases as the amount of force the material can experience decreases, and so does its cross-sectional area. The capacity of stress the material can experience decreases, and the strain the stress puts on the material increases. The R2 indicates the percentage of the dependent variable's variance that the independent variable can account for. R-squared, or the "goodness of fit," measures how well the data match the regression model. A perfect R2 of 1, indicates that all the points perfectly align on the line of best fit (Taylor, 2023). The R2 for Figures 14 and 16 is above 0.9, this result shows that the results match the line of best fit excellently.
The data for periods 4 to 7 show irregularity in the relationship between the atomic radius and Young’s modulus. The R2 for Figures 9, 11, and 13 is below 0.25, this result shows that the results do not show a direct correlation between the independent and dependent variables. As atomic radius shows a constant trend of decreasing across the periods (for the first half of the graph) as the number of protons increases, shielding/core electrons remain constant, increasing the effective nuclear charge. The attraction of the valence electrons to the nucleus increases across the period as more protons are added, causing the electrons to get closer to the nucleus and decreasing the atomic radius.
The increase and then decrease in Young’s modulus when going across periods 4 to 7 is because the atomic arrangements become more complicated, and the change from the s orbital to the p orbital is more extreme as the d orbital is skipped. The change accounts for a wider range of bonding types and crystal structures. The behavior of materials and their mechanical properties may exhibit irregularities or fluctuations due to these complexities. Young’s modulus trend is that it starts as a lower number, then increases and reaches a value lower than the first by the third element in the period. This trend is significant and not an anomaly because it is observed across all the periods from 4 to 7 as a continuum. The reason for this trend across the periods is, firstly, the change from alkali metals to alkali earth metals. Alkaline earth metals have two valence electrons, compared to one for alkali metals. In comparison to alkali metals, alkaline earth metals have higher Young's modulus values and greater stiffness due to the stronger metallic bonding interactions made possible by their extra valence electrons. Concerning the atomic radius, alkaline earth metals, which are more to the right of the period, have smaller atomic radii than alkali metals of the same period. Alkaline earth metals have more closely packed metallic structures due to their smaller atomic radii, which strengthen their metallic bonds and increase their Young's modulus values because of their greater resistance to deformation.
Moreover, when moving across periods 4 to 7 from groups 2 to 13, the elements approach the metalloids and non-metals, which affects the material's elasticity and stiffness. Compared to metals, metalloids usually have lower electrical and thermal conductivity. Their inclusion in a material may affect its conductivity, which, in turn, may alter how the material behaves under stress and deformation.
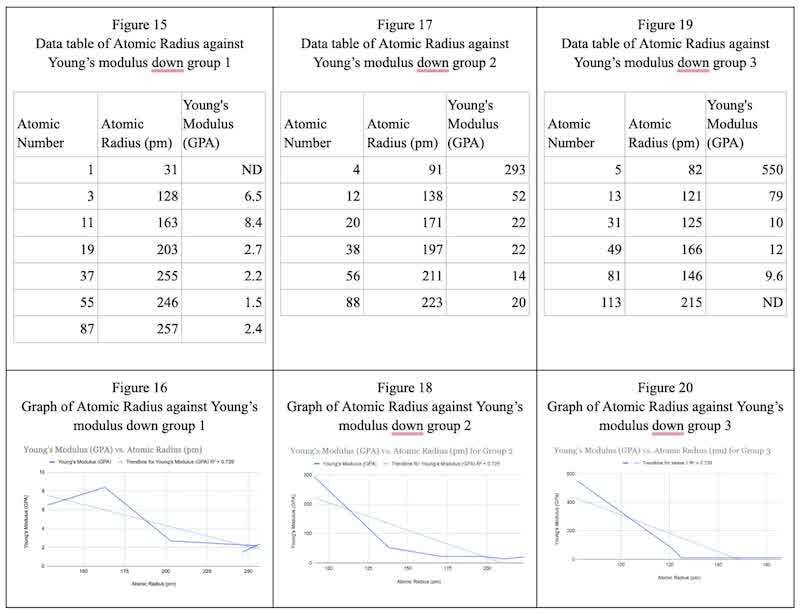
The data above show trends in atomic radius and Young’s modulus, spanning Figures 16, 18, and 20. Figure 16 is different from Figures 18 and 20 for the beginning section because, as supported by Figure 13, atomic number 3 shows a contrast in which Z=3 has a lower Young’s modulus than Z=11 but higher than Z=19, Z=37, Z=55, and Z=87. An explanation for this is that the smallest and lightest element in Group 1 is lithium (Z=3). Due to its smaller atomic size (Z=11) than that of sodium, lithium may have stronger metallic bonding because its nuclei are closer to its valence electrons. Compared to sodium, lithium may have a higher Young's modulus and greater stiffness due to its stronger bonding.
Furthermore, the trend of Young’s modulus showing a negative correlation with atomic radius is because the decrease in atomic radius is greatest with one element down. For example, in Figure 15 (group 1), considering the change in atomic radius from Z=4 to Z=12 is around 40pm compared to the decrease in atomic radius being 8 pm from Z=38 to Z=56. This trend is also observed in Figure 17 (group 2), where the change in atomic radius from Z=5 to Z=13 is 420 pm, while the change from Z=49 to Z=81 decreases by 2.4 pm. (The larger increase in atomic radius from period 2 to 3 compared to period 4 to 5 is because the energy levels converge as they get higher, hence, the distance decreases.) This explains and supports the negative correlation between atomic radius and Young’s modulus. For the elements lower down the group, the trend is also observed but at a less drastic level, as the increase in atomic radius is much less (as the energy levels begin to converge going down a group), and hence the increase in Young’s Modulus is less substantial but still present.
The reason for the negative correlation between atomic radius and Young’s modulus going down a group is that the addition of energy levels (shells) while moving down a group causes the atomic radius to increase. There is a larger gap between the nuclei and valence electrons in larger atomic sizes. Because of the decrease in effective nuclear charge due to more shielding and an increase in energy levels, electrons in the outer shell are less attracted to the nucleus. The increasing atomic radius usually makes metallic bonding in the heavier elements weaker. Lower Young's modulus and reduced stiffness are outcomes of weaker bonding. Furthermore, larger, more radially symmetric atoms tend to be more compressible. As atomic size increases down the group, the material's increased compressibility reduces its resistance to deformation, lowering its Young's modulus.
As the area/cross-section of the material increases due to an increase in atomic radius, the material can withstand a smaller force. Figure 21 shows the general trend of atomic radius and Young’s modulus. Down a group, the electrons in the outer shell of the element lose attraction to the nucleus as the effective nuclear charge decreases, the extent to which it can tolerate high force decreases. When electrons are strongly attracted to the nucleus, the nucleus can withstand greater pressure and stress without altering its strain. Generally, across a period, the atomic radius decreases as the electron affinity increases. Ergo, Young’s modulus increases as the elements become more compact and their ability to tolerate deformation and pressure increases.
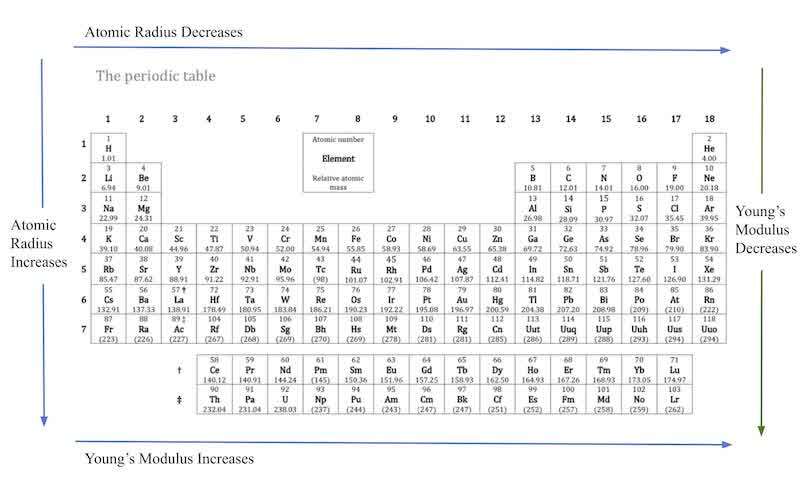
Figure 21: Periodic Table with Atomic Radius and Young’s Modulus Trends
The periodic relationship between atomic radius and Young’s modulus identified in this investigation has direct relevance to the formulation and performance of paints and protective coatings since paints function as a thin composite films whose durability, flexibility, and resistance to mechanical stress greatly depend on the mechanical properties of their inorganic components, including pigments and fillers (Boyondo et. al, 2025).
Materials composed of elements with smaller atomic radii tend to exhibit higher Young’s modulus values due to stronger nuclear attraction and tighter electron bonding. When used in pigments or extenders, such materials contribute to increased film stiffness, hardness, and scratch resistance (properties essential for industrial, automotive, and protective coatings). In contrast, components with larger atomic radii generally display lower Young’s modulus values, resulting in greater flexibility but reduced resistance to deformation. These materials are better suited to applications where elasticity and crack resistance are prioritized, such as exterior architectural paints exposed to thermal expansion and contraction.
Understanding how atomic-scale properties influence macroscopic mechanical behavior enables more informed material selection in paint chemistry. By leveraging periodic trends in Young’s modulus, formulators can better balance rigidity and flexibility within a coating system, thereby optimizing performance for the intended application. This demonstrates how fundamental atomic properties translate into practical considerations in materials engineering and industrial paint development.
Mariyah Matawala is Director of Business Planning and Analysis for Regent Paints and The Paint Foundation, where she helps contribute to zero-waste initiatives and promote circular economies by aligning projects with the organization’s mission and issuing certifications of excellence. Visit https://regentpaintsusa.com/ and http://thepaintfoundation.com/
References
- Aakash Educational Services Limited. (2023). Retrieved from https://www.aakash.ac.in/important-concepts/physics/young-s-modulus#:~:text=Value%20of%20Y%20 depends%20only,perfectly%20rigid%20body%20is%20infinite
- Britannica, T. Editors of Encyclopaedia (2023, November 10). Young’s modulus. Encyclopedia Britannica. https://www.britannica.com/science/Youngs-modulus
- Buyondo, A., Kasedde, H., Kirabira, J. B., & Yusuf, A. A. (2025). Integration of fillers in paint formulation: Comprehensive insights into methods, properties, and performance. Results in Engineering, 26, 105543. Retrieved from https://www.sciencedirect.com/science/article/pii/S2590123025016135?utm
- Garrett, J. (2023, November). Periodic Trends in the Periodic Table. IB HL Chemistry. New York: 1 Morris St, New York, NY 10004.
- Urone, P. P., Hinrichs, R., Dirks, K., & Sharma, M. (n.d.). 5.3 Elasticity - Stress & Strain. In Professor Emeritus (Ed.), College Physics by OpenStax (First Edition). XanEdu Publishing Inc.
- Retrieved from https://openstax.org/books/college-physics/pages/preface Wolfram Research, Inc. (n.d.-a). Covalent Radius of the elements. Wolfram Research, Inc. (n.d.-b). Young Modulus of the elements.



































