This study investigates the effects of modified formulations (particle size, aluminum flake addition) and inerting measures (oxygen reduction) on the explosion propagation of coating powder by using visual experiments.
We found a direct correlation between reduced particle size, the addition of aluminum flakes, and increased explosion risk. While reducing the oxygen content effectively slows flame propagation and combustion rate, its influence is modulated by particle size and composition. Notably, smaller particles exhibit slower flame propagation under low-oxygen conditions. The addition of aluminum flakes accelerates combustion, requiring a lower oxygen content for effective inerting.
Our comprehensive evaluation of combustion rates suggests an oxygen concentration below 15% for optimal inerting.
1. Introduction
As global regulations on volatile organic compound (VOC) emissions become increasingly stringent, low-VOC powder coatings, with outstanding environmental performance and excellent physicochemical properties, have found widespread application in industrial fields. In China, the implementation of strict source control mechanisms has accelerated the promotion of low-VOC powder coatings, driving the transformation of the coatings industry from liquid coatings to powder coatings. However, in order to further enhance the performance of powder coatings, such as smoothness, adhesion, conductivity, wear resistance, and corrosion resistance, researchers have actively developed new formulations. These innovations, however, may bring new hazards, specifically, an increased risk of dust explosions. For example, reducing the particle size increases the specific surface area and chemical reactivity of the dust, thereby increasing explosion sensitivity; the addition of metallic additives, especially aluminum powder, significantly raises the intensity of dust explosions and flame propagation speed due to its high reactivity. Therefore, with the growing use of powder coatings, the increase in dust explosion incidents has attracted significant attention from regulatory bodies and researchers. (1−9)
In recent years, researchers have conducted studies on the explosive characteristics of coating powders. For example, Choi et al. (10) studied the effect of aluminum powder content on the minimum ignition energy, finding that the high sensitivity of aluminum powder is closely related to particle size; Zhang et al. (11) studied the flame propagation characteristics of polypropylene powder dust explosion and found that the combustion process includes the diffusion combustion of large particles and the vapor phase combustion of combustible gases from the pyrolysis of small particles. Qin et al. (12) found that the explosion hazard of novel acrylic coating powders is greater than that of traditional epoxy and polyester coating powders. Zhang et al. (13) pointed out that the combustion of nanoscale PMMA dust exhibits macro characteristics of premixed gas combustion coupled with micro characteristics of localized diffusion flame combustion. These studies suggest that coating powders, due to their diverse components and complex combustion mechanisms (involving multiphase combustion), exhibit significant differences in explosion characteristics, which pose challenges for the risk assessment and prevention of dust explosions. Based on these studies, researchers have proposed several prevention and control measures, mainly focusing on inert gas suppression and chemical inhibition. For example, Choi et al. (14) effectively alleviated electrostatic dust explosions by increasing nitrogen concentrations. Pang et al. (15) found that carbon dioxide is more effective than nitrogen in suppressing the explosion intensity of low-density polyethylene dust. Cai et al. (16) pointed out that under high-pressure conditions, the inerting effect is limited, and further reduction of oxygen concentration is necessary for improved safety. Guo et al. (17) studied the inhibitory effect of NaHCO3 on the thermal degradation of PMMA dust and developed a kinetic model. These measures have been experimentally validated and proven effective, but in practical applications, factors such as cost control and the method of applying inerting substances have not been sufficiently considered, which affects their feasibility.
Meanwhile, during an investigation of an electrostatic powder coating line dust explosion accident, we found that the enterprise involved lacked awareness of the potential increase in explosion risks due to modified coating formulations and therefore had not implemented corresponding protective measures. To address this issue and achieve low-cost prevention, we worked with the enterprise to explore a solution: recycling combustion exhaust gases from the heating system on the coating line for inerting the coating dust, thereby reducing the higher dust explosion risks posed by the modified formulations. However, a key area that requires urgent study is the comprehensive effect of modified coating formulations and inerting protective measures on the explosion propagation mechanism, a topic that has not been reported in the literature so far. Therefore, this study aims to evaluate the practical viability of the proposed scheme and expand upon previous research in this field. We collected powder coating samples from the accident site and, using a visualized explosion propagation device, systematically investigated the combined effects of powder particle size, aluminum additives, and oxygen content in inert measurements on explosion characteristics and inerting efficiency. The findings of this research provide a theoretical basis for the development of more effective protective measures and offer strong support for safe production practices in the coatings industry.
2. Experimental Apparatus and Materials
2.1. Experimental Apparatus
The experiments were conducted in a rectangular explosion pipe (800 mm high, 80 mm × 80 mm cross section), as shown in Figure 1. The pipe was constructed with optical silicon glass on the front and back faces to allow for flame propagation visualization, and stainless steel sides. The bottom end was sealed with a stainless steel plate, while the center was equipped with a mushroom-shaped dust dispersion nozzle. A 0.12 mm thick PVC explosion vent diaphragm covered the top. Prior to each experiment, a precisely measured quantity of coating powder was evenly distributed around the nozzle, and the pipe was evacuated. Exhaust gases/air with varying oxygen content (0.45 MPa charging pressure) were then introduced via a 5 L distribution tank, with gas concentrations controlled by partial pressure matching. A solenoid valve was opened to inject high-pressure air through the nozzle, dispersing and mixing the powder to create a uniform dust cloud within the pipe. Continuous air injection increased the pipe pressure to near atmospheric levels. Dust concentration was calculated as the mass of the dust sample divided by the pipe volume. Ignition was initiated by a spark discharge between two tungsten–copper alloy electrodes positioned at the center of the pipe, 130 mm from the bottom. The energy of the ignition electrode is 20 J, and the ignition delay time is 250 ms after opening the solenoid valve. The flame propagation process was captured by a high-speed camera with a resolution of 1920 × 1080 pixels and a frame rate of 1000 frames per second. The PC-based synchronization system controls the trigger of the solenoid valve, the ignition system, and the high-speed camera.
Figure 1. Schematic diagram of the experimental apparatus.
2.2. Experimental Materials
A series of black sand textured coating powders used in the accident site were collected as the research object, including a coarse sand version (sample A), a fine sand plate version (sample B), and an aluminum flakes version (sample C). The formulation ratios of the three samples are basically the same, which is composed of about 23.80:31.50% epoxy resin/polyester resin, 32.10:4.50% inorganic filler (barium sulfate/titanium dioxide), 4.58% pigment, and 3.52% other additives. Table 1 details the key parameters of the three samples, including median particle size (D50), maximum explosion pressure (Pmax), lower explosive limit (LEL), and minimum ignition energy (MIE). Sample C demonstrates the highest explosion severity, with a Pmax exceeding that of Sample A and Sample B by 56 and 24%, respectively. Additionally, Sample C has the lowest LEL and the minimum MIE, indicating that it is the most easily ignitable among the three samples. This indicates that particle size reduction (without any observed agglomeration) and aluminum flakes addition are associated with the increase of explosion sensitivity, which is consistent with previous research (10) results. To investigate the effect of the oxygen concentration on dust explosions, an appropriate dust concentration must be selected for the experiment. Preliminary studies indicate that excessively high dust concentrations suppress explosions, while concentrations that are too low fail to sustain stable flame propagation. Therefore, to clearly observe the impact of the oxygen concentration and ensure the reliability of the experimental results, a dust concentration of 300 g/m3 was chosen as the standard value. This concentration effectively ensures explosion occurrence and allows for the examination of oxygen’s influence on flame propagation. At the same time, based on previous research results and considering the cost-effectiveness of future inerting measures, three oxygen contents of 21, 18, and 15% were selected as inerting conditions.
Table 1. Explosion Parameters of the Coating Powders
| type | D50 (μm) | LEL (g/m3) | MIE (mJ) | Pmax (MPa) |
| sample A | 68 | 75 | 90–100 | 0.43 |
| sample B | 35 | 75 | 35–45 | 0.54 |
| sample C | 38 | 25 | 15–25 | 0.67 |
3. Results and Discussion
3.1. Influence of Oxygen Content and Cross-Linking and Curing Reaction on Flame Propagation
To visually depict the flame propagation process, we present the flame morphology (Figure 2) and the variation in the flame propagation distance (Figure 3). While the speed curve can be derived from Figure 3 through differentiation, this approach is subject to errors due to limitations in numerical differentiation accuracy, equipment frame rate, and image resolution, making it challenging to correlate with the images at key moments in Figure 2. Therefore, we choose to qualitatively describe the variation in flame propagation speed using Figure 2, ensuring the reliability of the analysis and effectively capturing the dynamic characteristics of flame propagation. Because the three samples had different flame propagation speeds, the ignition moment was chosen as the start frame and the flame front reaching the sealing position at the top of the pipe was chosen as the end frame. The whole flame propagation process was shown over nine frames with equal time gaps (Figure 2).
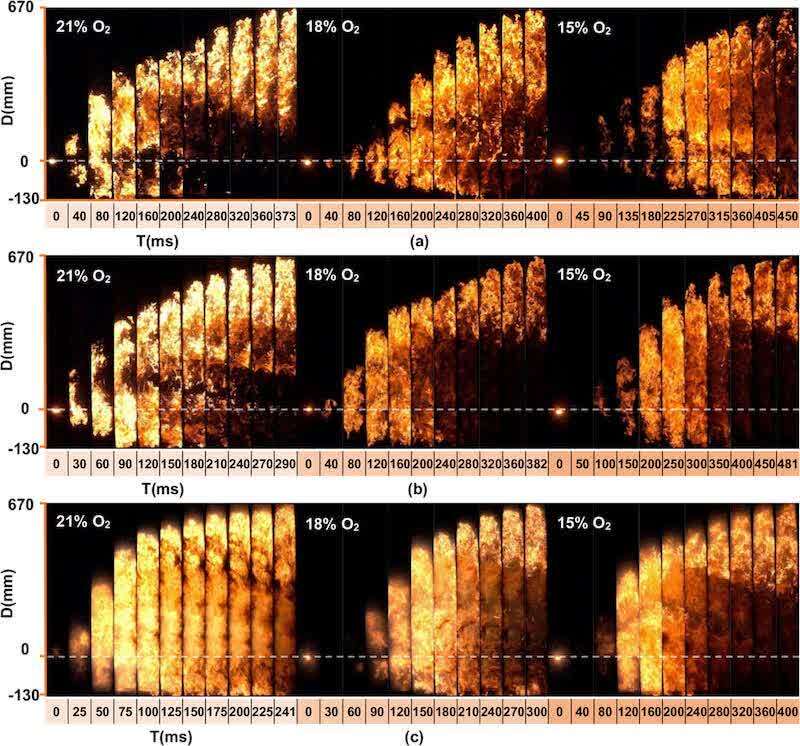
Figure 2. Flame propagation images of three samples at different oxygen levels: (a) sample A, (b) sample B, (c) sample C.
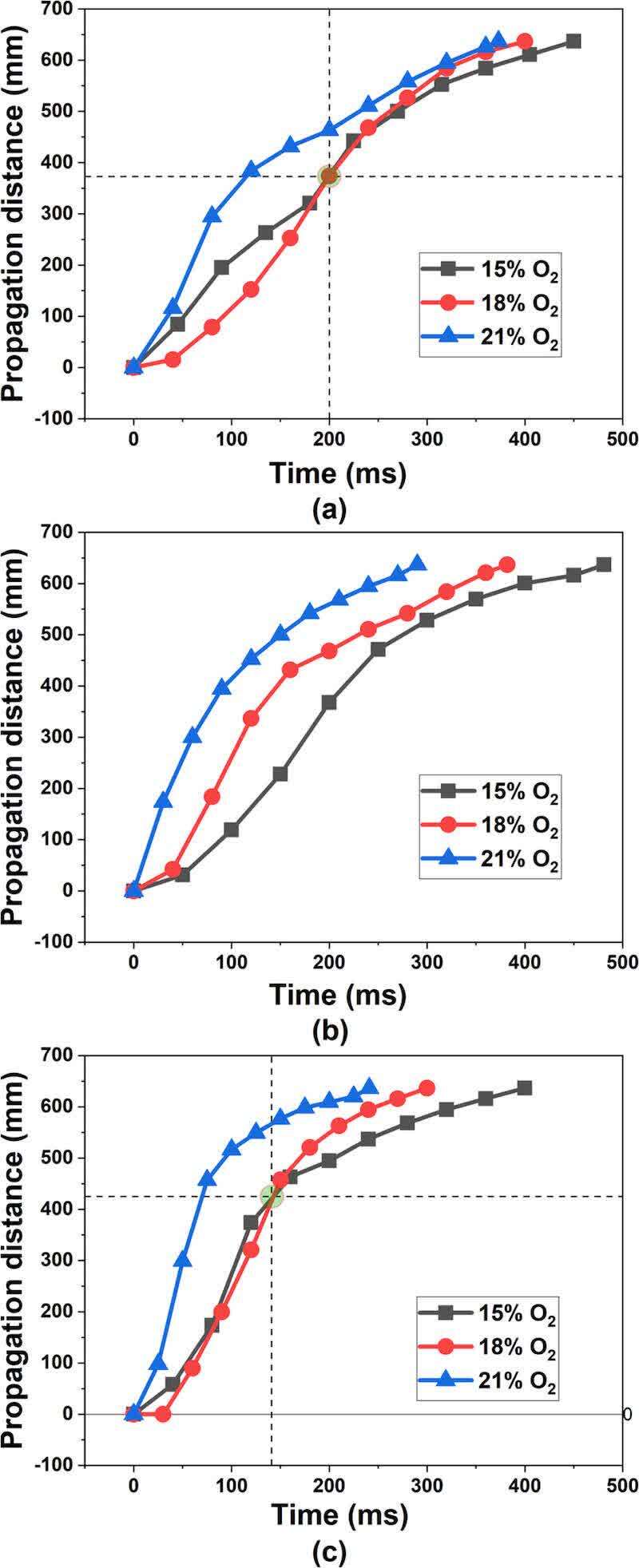
Figure 3. Flame propagation distance vs time: (a) sample A, (b) sample B, (c) sample C.
Figure 2 illustrates the impact of reduced oxygen concentration on the flame propagation time. A 3% reduction in oxygen concentration led to increased propagation times in all samples, with increases of 27 and 50 ms for Sample A, 92 and 99 ms for Sample B, and 59 and 100 ms for Sample C. This observed delay in propagation time suggests a nonlinear correlation between decreasing oxygen concentration and its suppressive effect on flame propagation. Furthermore, Figure 3 demonstrates that flame propagation velocities for all samples were highest at an oxygen concentration of 21%, decreasing with concentrations of 18 and 15%. Analysis of the flame propagation distance–time curves revealed an intersection point for Samples A and C at 375 and 425 mm, respectively, under 18 and 15% oxygen concentrations. This intersection indicates that the average flame propagation speed at 18% oxygen content surpassed that at 15% oxygen content beyond these points. The epoxy resin and polyester resin groups will cross-link reactively in the coating powder at the given heating temperature, curing to form a three-dimensional network structure after cooling. (18,19) Similarly, dust explosions elevate the temperature of the coating powder, triggering the same reaction that alters the powder’s physical and chemical properties and influences flame propagation. To elucidate the impact of this reaction on flame propagation, the initial flame propagation variations of Sample A were compared under 18 and 15% oxygen concentrations. Figure 4 illustrates that at 18% oxygen content, the post-ignition temperature increase causes adjacent powder to reach the cross-linking curing temperature. This forms a structure that hinders heat and oxygen exchange, slowing the pyrolysis rate. Consequently, the more pronounced cross-linking reaction at 18% oxygen content results in a lower flame propagation speed compared to 15% oxygen content. As the temperature further increases, this cross-linked structure breaks down, the powder partially decomposes, releasing a significant amount of gaseous combustibles, and both combustion intensity and flame propagation speed increase. Ultimately, flames with oxygen content exceeding 15% exhibit rapid expansion. Conversely, at 15% oxygen content, the initial combustion reaction is slower, releasing less heat. The flame ignites only a small amount of powder, and the discrete powder particles undergo pyrolysis and vaporization into the gas phase for combustion, hindering the formation of a continuous cross-linked structure. Due to the reduced obstruction from this initial cross-linking, the gaseous flame rises faster than at 18% oxygen content. However, downward propagation is slower until the combustion scale expands sufficiently to form cross-linked curing and establish a continuous combustion zone. Therefore, at 15% oxygen content, the initial flame appears sparse and less luminous, exhibiting a narrow and discontinuous distribution pattern.
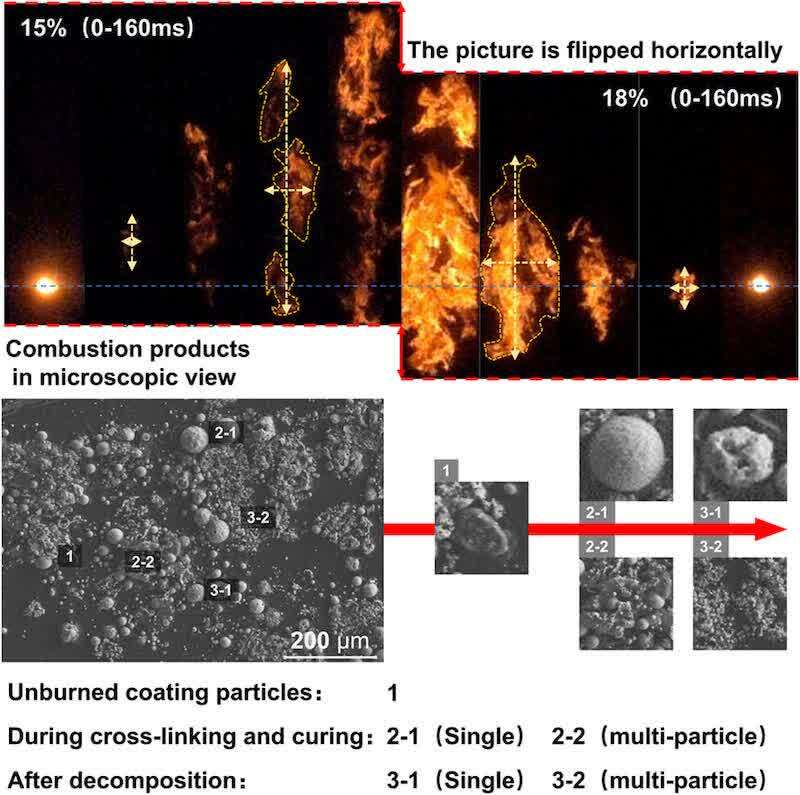
Figure 4. Impact of cross-linking curing on early flame propagation dynamics.
Figure 4 also shows scanning electron microscope images of the combustion products of Sample A. Partially pyrolyzed and combusted coating particles that did not undergo complete cross-linking and curing can be observed, forming individual spherical products. On the other hand, the interconnected porous combustion products indicate that some particles did experience cross-linking and curing reactions. These phenomena corroborate the inference from flame image analysis that cross-linking and curing might influence the initial flame propagation. Although the aforementioned phenomena were also present in Sample C, the difference in propagation speed between 15 and 18% oxygen content was not as pronounced as in Sample A. However, the average flame propagation speed of Sample B consistently decreased with decreasing oxygen content. It is preliminarily speculated that the addition of aluminum flakes and the reduction in particle size weakened the influence of cross-linking and curing on flame propagation. It can be affirmed that oxygen content, by affecting the cross-linking and curing reactions among the multiple components of the coating, consequently influenced the degree to which it hindered the flame burning rate, ultimately altering the evolutionary trend of the flame propagation velocity.
3.2. Effects of Oxygen Content on Flame Morphology and Combustion Rate
Figure 5 illustrates the final flame morphology for each sample at different oxygen levels (extracted from Figure 2). Sample A exhibits a gradual downward elongation of the combustion zone with decreasing oxygen content, indicating a progressively slower extinguishment of the tail flame as it propagates upward. In contrast, Sample B displays the opposite trend, with a faster extinguishment rate likely due to its smaller median particle size compared with Sample A. Consequently, the lower combustion zone of Sample B is fully extinguished by the time the flame front reaches the top of the pipe. Sample C retains more tail flame than Sample B, which can be attributed to its higher propagation speed. In this case, the lower combustion zone remains partially active when the flame front reaches the top.

Figure 5. Comparison of the flame propagation morphology in the end frame.
Due to limitations in our experimental setup, direct measurement of the flame combustion speed was not feasible. However, our comparative analysis suggests that a faster-extinguishing tail flame coupled with further flame front propagation indicates a higher burning speed. Therefore, we defined a quantitative index for combustion speed as the ratio of the flame front propagation distance to the time of initial tail flame extinguishment. Figure 6 presents a comparison of flame burning velocities under different conditions using this index. We observed that the combustion rates of Samples A and C decreased with decreasing oxygen content, with the rate of decrease diminishing gradually. At 15% oxygen content, the combustion rates of Samples A and B were similar, suggesting that at least 15% oxygen is necessary to mitigate the increased explosion risk associated with a reduced particle size. Interestingly, the combustion rate of Sample C at 15% oxygen content was comparable to that of Sample B at 18%, indicating that further oxygen reduction can effectively reduce the combustion rate of Sample C.
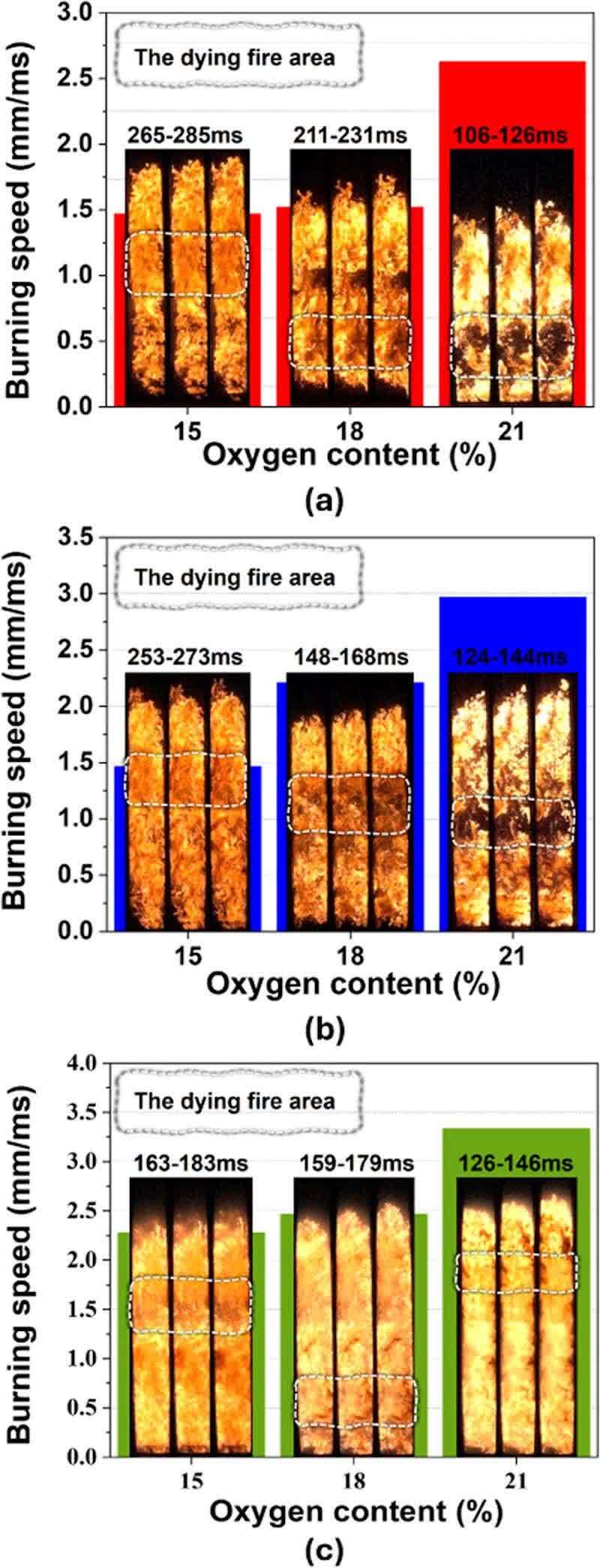
Figure 6. Quantitative comparison of combustion velocity: (a) sample A, (b) sample B, (c) sample C.
3.3. Influence of Coating Powder Formulations on Inerting Measures
As shown in Figure 2, at 21% oxygen content, Sample B’s flame reached the top of the pipe 83 ms earlier than Sample A’s. This lead time decreased to 18 ms at 18% oxygen. However, at 15% oxygen, Sample B lagged behind Sample A by 31 ms. This inconsistency suggests that the effect of particle size reduction on the flame propagation speed is not uniform under inerting conditions with reduced oxygen. Figure 7 highlights the differences in flame morphology between Samples A and B at two time points (200 and 280 ms) with 18% oxygen. At 200 ms, Sample B’s flame propagated further with a larger extinguished area, indicating faster initial burning. However, by 280 ms, Sample B’s combustion area decreased rapidly, while Sample A’s flame remained vigorous, with a propagation distance approaching that of Sample B. This demonstrates that Sample A exhibits higher combustion and propagation velocities in the later stages. This difference was even more pronounced at 15% oxygen. Our analysis of flame morphology at different time points suggests that smaller particles (Sample B) propagate faster under high oxygen conditions but experience weakened combustion in the later stages under low oxygen. This occurs because smaller particles burn more intensely, leading to rapid local oxygen depletion and faster extinguishment in low-oxygen environments. Therefore, reducing oxygen content ultimately reverses the impact of particle size on flame propagation speed.
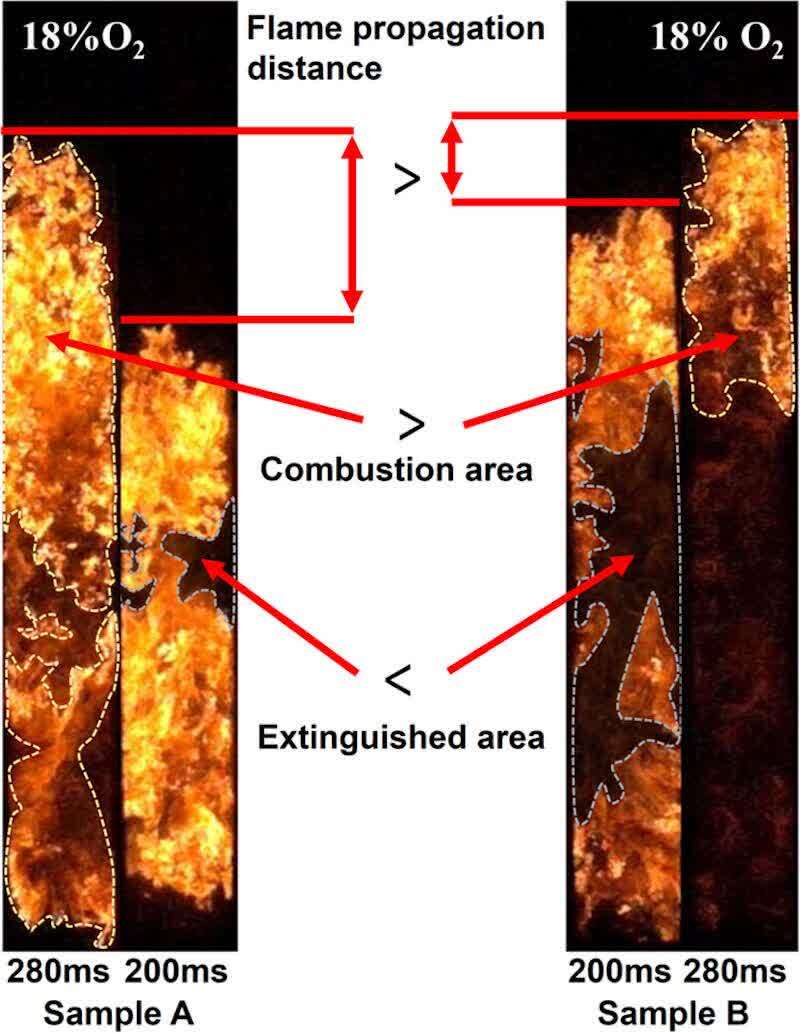
Figure 7. Comparison of flame morphology between samples A and B.
Figure 8 contrasts the leading edge region of the flame. Sample C shows more brightness at 18% oxygen concentration and 120 ms with a clear glowing zone at its edge. This implies that the combustion of Sample C generates greater heat radiation than Samples A and B. As a preheating zone for the explosion flame front, the glowing zone accelerates the pyrolysis process of the unburned powder coating. When the oxygen content was reduced to 15%, at the end of flame propagation (400 ms), there was a meteoroid phenomenon in the flame of sample C. This indicates that the coating powder is in the solid-phase combustion state of slow pyrolytic gasification. Under the action of gravity, a small amount of the gas phase burned away from the core combustion zone, forming a meteoroid flame trail.
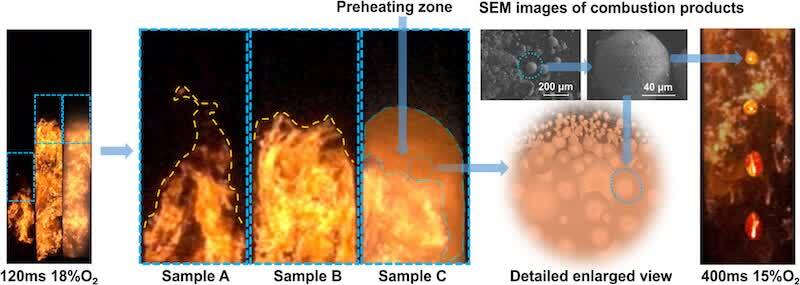
Figure 8. Influence of aluminum flakes on the propagation of explosion flames.
Based on the analysis of flame morphology at the two observed time points, the addition of aluminum flakes significantly enhances the combustion intensity of the coating dust, rapidly consumes oxygen, and accelerates the temperature rise. Therefore, coatings containing aluminum flakes must maintain a lower oxygen content to effectively achieve inerting. However, the current results do not clearly reveal the interaction mechanism between the combustion of aluminum flakes and the unique cross-linking and curing reactions of the coating dust, and no similar studies have been reported in the literature. Future research will further explore the underlying mechanisms of this phenomenon to overcome the limitations of the current study.
4. Conclusions
This research investigated the influence of modified formulations (particle size and aluminum flake addition) and inerting measures (oxygen concentration reduction) on the explosion propagation behavior of coating powders. Decreasing the particle size and incorporating aluminum flakes were found to increase the explosion risk. Reducing the oxygen concentration proves to be an effective inerting measure. However, this effect is influenced by the particle size and composition of the powder. Notably, fine powders exhibited slower flame propagation speed under low-oxygen conditions due to rapid oxygen depletion within the flame front. Conversely, the presence of aluminum flakes accelerated combustion, necessitating more stringent inerting conditions. An oxygen concentration below 15% is recommended for the effective inerting of these powders. Further research is needed to explore the complex interaction among aluminum flakes, cross-linking and curing reactions, and inerting mechanisms.
Haishun Xu is with the College of Mechanical and Electrical Engineering, China Jiliang University, Hangzhou 310018, China. Jie Mu is with the Zhejiang Academy of Emergency Management Science, Hangzhou 310012, China
Author Contributions: The article was written through contributions of all authors. All authors have given approval to the final version of the article.
Notes: The authors declare no competing financial interest.
Acknowledgments: This research was funded by the Zhejiang Provincial Natural Science Foundation of China (LQ20E040006) and the open topic of the key laboratory of the Ministry of Emergency Management (2020XFZB13).
References
- Wu, X.; Ning, B.; Li, X.; Deng, K.; Xu, P. 2022 Annual Report of China Powder Coatings Industry. Coat. Prot. 2024, 45 (2), 57−64.
- Powder Coatings and Painting Branch of CNCIA. Review of China Power Coatings Market in 2023 and Outlook of Development in 2024. China Coat. 2024, No. 4, 14−17.
- Du, Z.; Wen, S.; Wang, J.; Yin, C.; Yu, D.; Luo, J. The Review of Powder Coatings. J. Mater. Sci. Chem. Eng. 2016, 04 (3), 54−59.
- Ayrilmis, N. A Review on Electrostatic Powder Coatings for the Furniture Industry. Int. J. Adhes. Adhes. 2022, 113, No. 103062.
- Farshchi, N.; Gedan-Smolka, M. Polyurethane Powder Coatings: A Review of Composition and Characterization. Ind. Eng. Chem. Res. 2020, 59 (34), 15121−15132.
- Li, G.; Yuan, C.; Chen, B. Explosion Risk Evaluation During Production of Coating Powder. J. Hazard. Mater. 2007, 149 (2), 515−517.
- Yang, K.; Zhai, M.; Pang, L.; Zhang, Y. Research on Risk Assessment Method of Dust Explosion in Electrostatic Powder Coating Firms. J. Loss Prev. Process Ind. 2023, 86, No. 105198.
- Bielawski, J. Fire and Explosion Hazards Associated with Electrostatic Powder Coating. Zesz. Nauk. SGSP 2020, 75, 23−36.
- Eckhoff, R. K.; Pedersen, G. H.; Arvidsson, T. Ignitability and Explosibility of Polyester/Epoxy Resins for Electrostatic Powder Coating. J. Hazard. Mater. 1988, 19 (1), 1−16.
- Choi, K.; Sakurai, N.; Yanagida, K.; Itoh, H. Ignitability of Aluminous Coating Powders Due to Electrostatic Spark. J. Loss Prev. Process Ind. 2010, 23 (1), 183−185.
- Zhang, S.; Yan, X.; Wen, Z.; Lu, Y.; Shi, S. Study on Flame Propagation Characteristic and Mechanism of Polypropylene Dust Explosion. Powder Technol. 2024, 440, No. 119794.
- Qin, X.; Zhang, Y.; Shi, J.; Wei, X. Study on Explosion Characteristics and Mechanism of Electrostatic Spray Powder. ACS Omega 2024, 9 (17), 19645−19656.
- Zhang, X.; Gao, W.; Yu, J.; Zhang, Y.; Chen, H.; Huang, X. Flame Propagation Mechanism of Nano-Scale PMMA Dust Explosion. Powder Technol. 2020, 363, 207−217.
- Choi, K.; Choi, K.; Nishimura, K. Experimental Study on the Influence of the Nitrogen Concentration in the Air on the Minimum Ignition Energies of Combustible Powders Due to Electrostatic Discharges. J. Loss Prev. Process Ind. 2015, 34, 163−166.
- Pang, L.; Zhao, Y.; Yang, K.; Zhai, H.; Lv, P.; Sun, S. Law of Variation for Low Density Polyethylene Dust Explosion with Different Inert Gases. J. Loss Prev. Process Ind. 2019, 58, 42−50.
- Tsai, Y. T.; Ho, S. C.; Huang, A. C.; Shu, C. M. Potential Explosion Hazard of Polyester Resin Dust Formed from a Granulation Process: Limiting Oxygen Concentration with Different Pressures. Appl. Therm. Eng. 2018, 135, 74−82.
- Guo, R.; Li, N.; Zhang, X.; Zhang, Y.; Chen, Q.; Zhao, W.; Wang, X.; Li, R. Suppression Mechanism of Micron/Nano PMMA Dust Flame Based on Thermal Analysis. Adv. Powder Technol. 2022, 33 (12), No. 103848.
- Gedan-Smolka, M.; Lehmann, D.; Cetin, S. Basic Investigations for Development of New Curing Mechanisms for Powder Coatings. Prog. Org. Coat. 1998, 33 (3−4), 177−185.
- Czachor-Jadacka, D.; Pilch-Pitera, B.; Kisiel, M.; Thomas, J. Polyurethane Powder Coatings with Low Curing Temperature: Research on the Effect of Chemical Structure of Crosslinking Agent on the Properties of Coatings. Prog. Org. Coat. 2023, 182, No. 107662. 6649



































