Wastewater treatment in metal finishing processes always encompasses critical procedures.
 Stephen Rudy, CEFThus, it is the singular most important part of the metal finishing operation. Being out of compliance results in fines, more frequent municipal and state testing, and the presentation of an operations plan amendment to show cause for improving the waste treatment system. The ultimate in violations is operating shut down. This, in turn, shuts down the metal finishing plant processing, a most crippling and costly situation. For job shops, once their customers find another finishing source, those customers may not return when the system is back in operation. Captive shops would need to outsource their work, resulting in a negative hit to the profit margin.
Stephen Rudy, CEFThus, it is the singular most important part of the metal finishing operation. Being out of compliance results in fines, more frequent municipal and state testing, and the presentation of an operations plan amendment to show cause for improving the waste treatment system. The ultimate in violations is operating shut down. This, in turn, shuts down the metal finishing plant processing, a most crippling and costly situation. For job shops, once their customers find another finishing source, those customers may not return when the system is back in operation. Captive shops would need to outsource their work, resulting in a negative hit to the profit margin.
Critical steps in waste treatment include: maintaining correct pH at specific stages of treatment, oxidizing cyanides, reducing hexavalent chromium, precipitating metals, and removing oils, greases, and related organic materials. The primary driving force is compliance with federal, state, and municipal regulations. Meeting or exceeding discharge and effluent limitations daily almost ensures the facility will operate without fines or potential shutdown. Some installations opt to have a certified waste hauler remove and properly treat their wastewater. Others may operate without a sewer permit, whereas there is no discharge. This can incorporate the use of the waste hauler or purification technology, such as ion exchange or membrane systems. Many operations elect to treat their process waters in-house. These shops have a system in place to process water in specific steps, under a set of controlled operating procedures. The benchmark to maintain a viable on-site waste treatment system starts at 30,000-40,000 gallons daily discharge of treated water.
Typical Operation
A typical operation consists of:
- Collecting wastewater into an equalization tank. Various source plant waste waters are thoroughly mixed, producing a uniform blend ready for subsequent treatment.
- pH adjustment to range 7-9, by acid or caustic dosing (typically a dilute solution of either sulfuric acid or sodium hydroxide). Coagulant is introduced to initiate metal precipitation. It initiates charge neutralization, destabilizing the colloid, conditioning precipitants to settle densely.
- For metal finishing waste solutions, typically an anionic polymer is introduced to agglomerate solid metal hydroxides into larger particles, thereby accelerating settling.
- Treated water passes through a clarifier, further settling solids.
- Processed water may pass through a carbon filter or similar unit, such as a sand filter. This step polishes the water to excellent clarity.
- Collected, thickened sludge passes through a dewatering press and is packaged in T-Sacks or Geobags, in preparation for compliant shipping and disposal.
To effectively determine the addition rates of coagulant and polymer, a manual or automated jar testing unit is recommended. This method can set flow rates for both flow-through and batch systems.

Lab Type Jar Tester
Additional Steps to Accommodate Requirements
Some treatment systems may require additional steps to accommodate specific requirements. One is the two-step cyanide oxidation using sodium hypochlorite (industrial-strength bleach), typically 10-15% by volume. Another is sodium metabisulfite or ferrous sulfate, used to reduce hexavalent chromium to trivalent chromium.
The two-step cyanide oxidation is accomplished in two separate tanks. In the first tank, caustic (liquid sodium hydroxide) is added to achieve a pH range of 11-11.5. Then, the bleach solution is added to initiate the chemical conversion of cyanide to cyanate. Most installations use a calibrated ORP probe to automatically dispense the bleach solution to maintain the ORP range of 300-350 millivolts, ensuring complete phase 1 oxidation. In the second tank, additional bleach and dilute sulfuric acid are added to maintain a pH of 8-8.5. -Once the pH range is achieved, the same strength bleach solution is added to reach 600-650 millivolts. Sufficient holding time is required, approximately. 45 minutes to complete each of the two-step reactions. Completion of the second step results in the formation of nitrogen and carbon dioxide. Once the second-step reaction is complete, this treated solution can be combined with the previously segregated wastewater for basic treatment, as described in steps 2-6 above.
Hexavalent chromium reduction is accomplished by first adjusting the solution pH to 2-2.5 by adding dilute sulfuric acid. Then, a Sodium Metabisulfite solution at approximately 30% by weight is added as the reducing agent. The reaction is monitored by oxidation-reduction potential. The reducing agent, aqueous Sodium Metabisulfite, is metered in by an ORP probe interfaced with the dosing pump. The reduction of hexavalent chromium to trivalent chromium is complete when the stable ORP of 280 millivolts is achieved. Once the second-step reaction is complete, this solution can be combined with the previously segregated wastewater for basic treatment, as described in steps 2-6 above.
Some metal finishing processes include chelated systems, such as electroless nickel, alkaline copper, descalers, and burnishing. The problem occurs when these chelates are chemically strongly bound to metals. The conventional waste treatment process may have little to no effect on breaking these strong bonds. Precipitants are added with excellent effect to break the chelate-to-metal bond. Polysulfides and thiocarbamates are typically used in this step, typically by ORP, just before or in conjunction with the coagulant.
Depending on the process baths in the metal finishing operation, foam can be an issue during waste treatment. Cleaners and mass finishing compounds are examples of highly foaming solutions. Defoamers, silicone or non-silicone, are used to rapidly knock down objectionable foam.
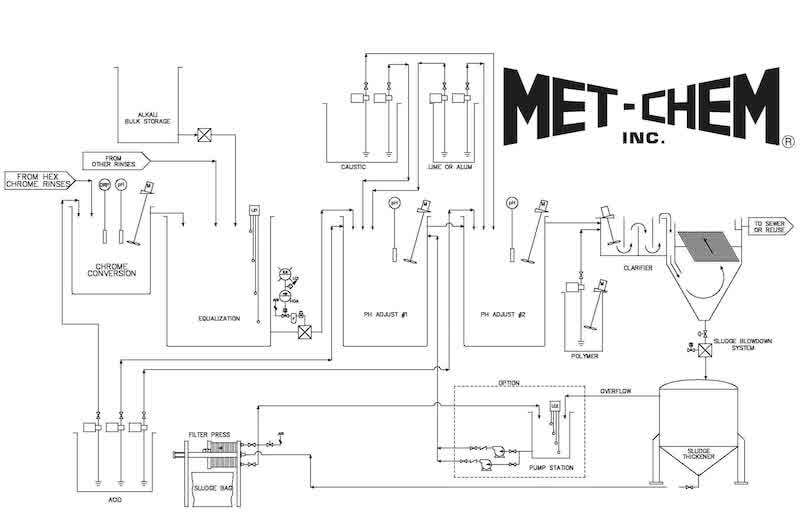
Schematic of a typical metal finishing waste treatment system.
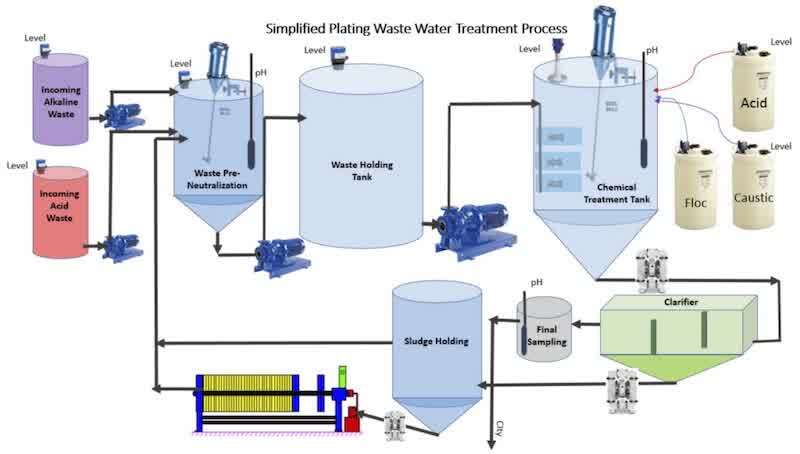
Individual additives metering into the waste treatment system.
Good Maintenance Practice
 UV/ViS Spectrophotometer.The waste treatment system, operating under proper conditions and steps, will effectively precipitate metals. These may also be referred to as heavy metals (e.g., chrome, nickel, iron, copper, zinc, silver, tin). Good maintenance practice includes cleaning pH probes and calibrating daily, checking dosing pumps to confirm the desired feed rate is achieved, and coordinating with the metal finishing line to compensate for tank dumps and new make-ups. Inventory of commodity and proprietary chemistries should never be down to “the last drum or tote.”
UV/ViS Spectrophotometer.The waste treatment system, operating under proper conditions and steps, will effectively precipitate metals. These may also be referred to as heavy metals (e.g., chrome, nickel, iron, copper, zinc, silver, tin). Good maintenance practice includes cleaning pH probes and calibrating daily, checking dosing pumps to confirm the desired feed rate is achieved, and coordinating with the metal finishing line to compensate for tank dumps and new make-ups. Inventory of commodity and proprietary chemistries should never be down to “the last drum or tote.”
Ample quantities must be on hand to address unexpected operating challenges and product sourcing lead times. Many facilities must submit samples of their treated water to a local or state-approved lab for analysis, typically monthly. It is necessary to regularly analyze discharge water in-house using commercially available instrumentation and test kits, especially when mandated water samples are to be tested by the certified lab.
Atomic Absorption Spectrophotometer.
This keeps the waste treatment system optimized and avoids preventable out-of-compliance fines.
Stephen F. Rudy, CEF, is president of Chem Analytic and has written extensively about the finishing industry. Visit www.chemanalytic.com or call him at 917-604-5001.




































