The porous pot was designed to purify hard chrome plating baths by supposedly removing iron, copper, and other tramp metals.
 Eric Svenson Sr.Some plating operations also use these to lower trivalent chrome levels in their baths. Porous pots are claimed to purify any solution that contains chromic acid. However, the practicality of using these for bath purification is a fallacy.
Eric Svenson Sr.Some plating operations also use these to lower trivalent chrome levels in their baths. Porous pots are claimed to purify any solution that contains chromic acid. However, the practicality of using these for bath purification is a fallacy.
Removing impurities is important because they tend to build up over time, causing problems with plating quality, production efficiency, and overhead costs. Hard chrome platers, in particular, have been struggling for decades with the effects of high levels of metallic impurities. The porous pot sounds like the magic wand for solving their problem. The reality, however, is the exact opposite because these actually do more harm than good.
There were several producers of these pots years ago, each having a slightly different design. Currently, only one manufacturer remains known.
The Design and Intended Mechanism
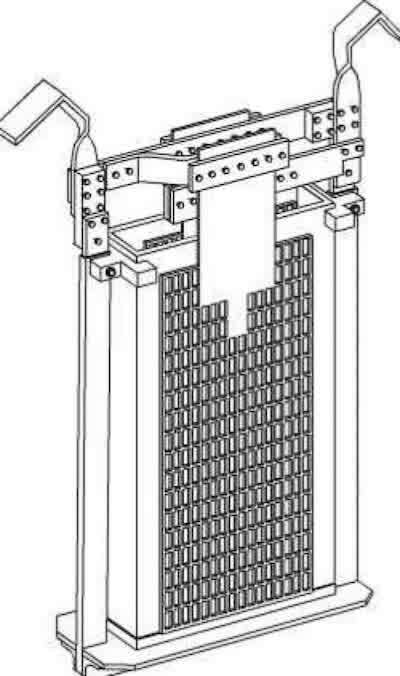 Schematic of a typical Porous Pot.The typical design consists of a porous ceramic open-top chamber, about 40% porous, with a 1- or 2-micron pore size. Compare that to a human hair, which is about 70 microns thick; the porous pots’ pore size is actually closer to that of a single bacterial cell.
Schematic of a typical Porous Pot.The typical design consists of a porous ceramic open-top chamber, about 40% porous, with a 1- or 2-micron pore size. Compare that to a human hair, which is about 70 microns thick; the porous pots’ pore size is actually closer to that of a single bacterial cell.
The pot’s top has two hooks, one positive and one negative, that connect to the tank’s bus bars. Dual anodes are located on the outside, and a single cathode is positioned in the pot’s center. These come in several sizes to accommodate various tank dimensions. Smaller tanks often use one pot while larger ones may use several pots. These have been run overnight when the tank is empty of work, or simultaneously during plating in an unused portion of the tank.
Before starting, the pot is filled with either the chrome bath or a sulfuric acid solution. However, using sulfuric acid could lead to mixing with the bath, thereby upsetting its ratio.
These supposedly operate by using DC to drive impurities in the bath through the ceramic pores via electromigration, where they become trapped and precipitate inside the pot. This action is said to separate the impurities from the bath, thereby purifying it.
In theory, the porous pot works; in reality, it doesn’t. The concept is based on the hydrogen that’s evolved at the cathode inside the pot. This releases hydroxide ions (OH-) from the electrolysis of water, increasing the alkalinity in the pot to a pH above 10. At that higher pH, almost all metals precipitate, including any chrome, iron, and copper. That’s the same basic method used to waste treat a chromic acid solution.
What Really Happens
The mentioned pH increase might help purify the bath if there were actual flow into and out of the ceramic pot. In reality, the pot’s pores get plugged up very quickly with minute particles of bath debris. This prevents any bath migration. Think about it; how much would the pot leak if it were filled with water and hung in the air? Barely any; the best you might get is some very slow weeping. That’s one reason the porous pot doesn’t work; the chrome bath doesn’t actually flow through it.
The whole concept is based on reducing the pH inside the pot. But there’s no bath flow through the pot when the pores plug, so the bath itself never gets treated. The only solution that’s treated is the pot’s initial filling. On the other hand, if there were adequate flow through the pot’s pores, then there wouldn’t be any pH change inside the pot and, therefore, no impurity removal.
Either way, with or without flow, the bath doesn’t get purified.
The Pot’s Precipitate
What actually happens inside the pot is that the increase in pH precipitates all the metals as hydroxides. This includes all of the usable chromic acid and any iron and copper that’s present. The hexavalent chrome inside the pot gets converted to trivalent chrome hydroxide (Cr(OH)3) and precipitates at the high pH. That’s the green sludge that builds up in the pot, which is so messy to remove.
What the porous pot really did was convert the good, usable chromic acid in the bath into hazardous waste. The user mistakenly believes they are purifying the bath when they are actually destroying good usable chromic acid by converting it into a waste product, a hazardous waste to boot. On top of that, cleaning out the green sludge is an incredibly messy operation.
Laboratory Testing
This was confirmed by analytical testing of a 1,225-gallon hard chrome bath. It contained 34 oz/gallon of chromic acid with a 98:1 sulfate ratio, 2.5% trivalent chrome, 6.8 g/l of iron, and 4.6 g/l of copper. The porous pot was filled with the bath and allowed to run in the tank for 48 hours at 6.5 Volts. There was no work being plated during that time, so no new impurities were introduced.
The sludge from inside the pot was then tested and found to contain the same percentages of chrome, iron, and copper as those in the bath. The vast majority of the pot’s sludge was found to be green chrome hydroxide, previously derived from usable chromic acid.
The bath was then tested and found to contain the same amounts of trivalent iron and copper as before the test began. Those impurities hadn’t been reduced at all. The only thing accomplished was wasting good chrome, some electricity, and generating a waste byproduct.
The takeaway is that using porous pots is counterproductive. They destroy good chromic acid and create excess hazardous waste.
A Better Approach
No question operating a chrome bath with high impurity levels causes various plating problems. These include pitting, poor throwing power, burning and dullness, slower plating speeds, higher electrical consumption, and increased rework rates.
If using porous pots isn’t the answer, then the question becomes, what’s the best way to purify a chrome bath? Two other off-the-shelf techniques are available, but both are also impractical:
- Electrodialysis: Electrodialysis can be made to work by separating the impurities from the chrome bath. The equipment needed, however, is very expensive, takes up significant floor space, and becomes a maintenance nightmare. The electrolytes used in the process need to be monitored and replaced frequently, and the costly membranes require regular replacement. As a result, most of the companies that promoted these systems are now out of business.
- Ion Exchange: Ion exchange systems can also be used to remove the bath’s impurities. However, the equipment is both expensive to purchase and costly to maintain. This process has three significant problems. The first is that the bath needs to be diluted with water to 50% before processing to extend the resin’s life. This requires evaporating the excess water before it can be returned to the plating tank. The second problem is that the resin has a limited lifespan, is expensive, and needs replacement at frequencies that depend on how it’s used. The third, and possibly the worst problem, is that the regenerates used to backwash the resin bed become a hazardous waste product. Disposing of this large amount of waste adds to the already high operating cost.
So, what’s the best way to purify a hard chrome bath that’s already contaminated? Well, that depends on what the impurity is.
Trivalent and Chloride
These are easily dummied out of the solution by using a properly designed dummy call after making a bath addition of CR-3 Reducer. When using the desired anode ratio, the trivalent form is converted back to usable chromic acid, while any chlorides present evolve at the anode surface. Details on this procedure were presented in an earlier article or are available by contacting the email shown below.
Heavy Metals like Iron and Copper
Sadly, these are difficult and more costly to remove. As indicated, using porous pots, electrodialysis, or ion exchange isn’t the answer. Unfortunately, there is no practical, off-the-shelf method for removing metallic ions due to the complex chemistry involved.
But, dumping and replacing a bath with high metallic impurities isn’t needed. Realize that no chrome bath is truly pure, because even a freshly made bath contains trace impurities from the water and the chemicals used. Moreover, there’s no adverse effect when these impurities are kept below the threshold at which they start causing problems.
Why the TCL Range is Important
TCL stands for Total Contaminant Loading and is simply the sum of the trivalent percentage plus the g/l of iron and copper present in the bath. As an example, a bath with 4% trivalent, along with 3 g/l of iron and 5 g/l of copper, would have a TCL of 12. Using TCL is an easy and accurate way to evaluate the bath’s condition.
A hard chrome bath with TCL below 4.8 will have little to no adverse effect on plating quality for most parts, so there’s no practical need to purify it below that level. Plating issues typically become a problem when the 7.2 TCL level is reached. Therefore, maintaining the bath within the 4.8–7.2 TCL range makes good business sense from both cost and quality standpoints.
The Practical Purification Method
The best and least expensive method for maintaining the TCL within the desired range is by decanting the bath. This is where a portion of the bath is pumped out to the 4.8 TCL level, and the portion removed is replaced with fresh solution. This dilutes the TCL down to a suitable operating level.
Bath decanting has proven to be the least expensive and most effective method of removing excess impurities.
Any excess trivalent present should be dummied out before decanting because this reduces the gallons that must be removed and lowers the expense. The cost of decanting typically runs around $25.00 per bath gallon, covering hazardous waste disposal and the chemicals needed to rebuild it. Yes, that’s costly, but consider it a sound investment because the plating bath is the lifeblood of your business. When it’s sick and out of balance, the business suffers from poor plating quality, increased rework, and higher costs.
The initial purification decant will be the costliest, but the rewards are well worth the expense. Future treatments to maintain the TCL within the desired range (mini-decants) will be less expensive because a smaller volume will need to be removed.
Decanting for bath purification can be done quickly, so you are back in business without much delay. The gallons of bath to be removed and the re-mixing chemicals needed can be calculated for you based on a recent bath analysis.
 On the left, the contaminated bath is darker. On the right, a pure bath appears redder.
On the left, the contaminated bath is darker. On the right, a pure bath appears redder.
Maintaining a Purified Bath
The bath should be operated to reduce impurity loading once it is initially decanted. Some of the more important considerations include:
- Dummy it using the CR-3 Reducer whenever the trivalent level exceeds 2%.
- If possible, reverse-etch in a separate etch tank to avoid introducing iron.
- Avoid using copper for the fixtures or contacts.
- Be careful not to introduce copper when cleaning the bus bars.
- Stop off all fixture areas under solution except the electrical contact points.
- Never use steel anodes and keep the lead alloy anodes active using AnoBest-104.
- Prevent metal filings from parts from being added and retrieve dropped items quickly.
Final Thoughts
Maintaining a relatively pure hard chrome bath is important from the standpoints of deposit quality, production throughput, and operational costs. The plating industry has recognized this for decades, but often uses the wrong approach. Porous pots don’t work and actually run counter to the desired outcome. They don’t remove iron or copper; instead, they waste electricity and tank space, destroy good chromic acid, and increase hazardous waste. The most effective, fastest, and least costly way to remove these contaminants is the old-school method of bath decanting. Yes, sometimes older is better, and this is one of them.
Aged, tired, and contaminated baths should be decanted to reduce impurity levels, then maintained to keep the TCL within the desired range. This practice results in the best chrome deposit quality, the highest production efficiency, and the lowest production costs
Eric Svenson Sr. is CEO of Plating Resources and a Master CEF and IUSF member. Visit www.Plating.com. If you have any questions or wish to explore this topic further, please contact Vicky at Vicky@Plating.com.




































