The purpose of using electroplating is to reduce the high cost of corrosion of metal equipment. However, the dangers of this process are the basis for using industrial hygiene control methods in this industry.
 Parnian Ilbag, Masoud Rismanchian, and Saeid Yazdanirad.The aim of this study is to investigate the effective parameters in the performance of the balls covering the surface of the plating bath on the amount of vapors that are released. This study was done in chrome plating. The balls covering the bath surface and the parts to be plated, which were the balls in gas ball valves used in this study, have two different outer diameters, the number of layers covered by plastic balls, and other parameters such as the temperature of the bath, the concentration of the solution inside the bath, and the humidity and temperature of the environment. In this study, each stage of the research includes 4 samples, and a total of 80 air samples were collected based on 7300 NIOSH analytical methods.
Parnian Ilbag, Masoud Rismanchian, and Saeid Yazdanirad.The aim of this study is to investigate the effective parameters in the performance of the balls covering the surface of the plating bath on the amount of vapors that are released. This study was done in chrome plating. The balls covering the bath surface and the parts to be plated, which were the balls in gas ball valves used in this study, have two different outer diameters, the number of layers covered by plastic balls, and other parameters such as the temperature of the bath, the concentration of the solution inside the bath, and the humidity and temperature of the environment. In this study, each stage of the research includes 4 samples, and a total of 80 air samples were collected based on 7300 NIOSH analytical methods.
Based on the results, the mean ± standard deviation of pollution concentrations without and with balls were 41.66 ± 59.99 mg/m3 and 26.67 ± 53.07 mg/m3. The results showed that the use of balls covering the surface of the plating bath could significantly reduce the pollution concentration (P = 0.004). However, parameters of balls in gas ball valves, number of ball layers, and number of plastic balls could not affect the pollutant emission with balls (P > 0.074). Therefore, it can be stated that the plastic balls covering the surface of the electroplating bath are an effective way to reduce the amount of pollutants emitted from the surface of the electroplating bath.
Introduction
Today, the use of all kinds of metal equipment is inevitable, and this equipment is the pillar of our civilization. Metal materials have a limited life due to exposure to external factors and environmental conditions [1]. The annual cost of metal equipment corrosion is estimated to be 3.4% of the global Gross Domestic Product (GDP) [2]. Therefore, both in terms of cost savings and the amount of material consumption, there is a need to adopt methods to maximize the life of the equipment. One of the ways to increase the life of metal equipment is to use the electroplating process. The electroplating process means covering the surface of one metal with a thin layer of another metal by electric current in a chemical solution [3]. In all stages of the electroplating process, various chemicals are used, and the metal materials include nickel, chrome, gold, zinc, etc. [4]. In the meantime, some substances such as nickel, chromium, and cadmium compounds are known as human carcinogens [5, 6], and therefore, the exposure of the workers to these chemical agents is one of the dangers in these industries and requires professional industrial hygiene interventions [4].
Scientific texts refer to methods to control pollutants in the electroplating industry. Each of the methods provides a certain level of control for the operator. The primary approach in all control steps is first to eliminate risks and, if that is not possible, to minimize the risk [7].
However, the stage of the process (before starting or during the process), the type of chemicals used in the electroplating process, and the implementation costs are the limiting factors in the selection of control methods [4].
The engineering controls used in this process include local ventilation, the use of surfactants or other solutions that increase the surface tension of the solution, the use of bubble dispersants on the surface of the solution, and the use of foam or plastic balls on the surface of the solution [8, 9]. Although removing the hazards and replacing the hazardous substance with less hazardous substances is the most desirable control method, it is not easily possible for all processes [9]. Also, the creation of local ventilation is costly, and the use of surfactants that increase the surface tension of the bath is also prohibited in some of its types due to secondary effects on the environment, and the types that are not harmful to the environment are expensive [4]. Using the foams covering the surface of the bath can lead to a reduction in pollutant production, but due to the trapping of oxygen and hydrogen produced in the baths, there is a possibility of these foams exploding. Although it is possible to reduce these risks by using foams with special compounds and the size of special openings, access to this type of foam is difficult and expensive [9]. Plastic balls covering the surface are one of the most popular methods for reducing surface evaporation, and their use to reduce surface evaporation from the surface of lakes has attracted the attention of researchers today. In the studies conducted in this field, the performance of these balls in reducing surface evaporation is estimated to be 20 to 40 percent. However, the secondary effects of producing and releasing these balls are one of the challenges of using them on the surface of lakes [10-12].
Therefore, it seems that the use of plastic balls covering the surface of the electroplating bath can be an effective way to reduce the exposure of operators to these pollutants. In this study, the effect of the balls covering the surface of the electroplating bath on the amount of vapors released from the surface of the bath and the determination of effective parameters in their performance in one of the electroplating workshops of BGas ball Valves Manufacturing Company in Isfahan province of Iran, are investigated.
Materials and Methods
Gas valve electroplating process
This process, which has two separate body and ball electroplating parts, includes two types of nickel and chrome plating. The process of plating the balls is shown in Figure 1. Also, the schematic view of the chrome plating bath as the main part of the study is shown in Figure 2.
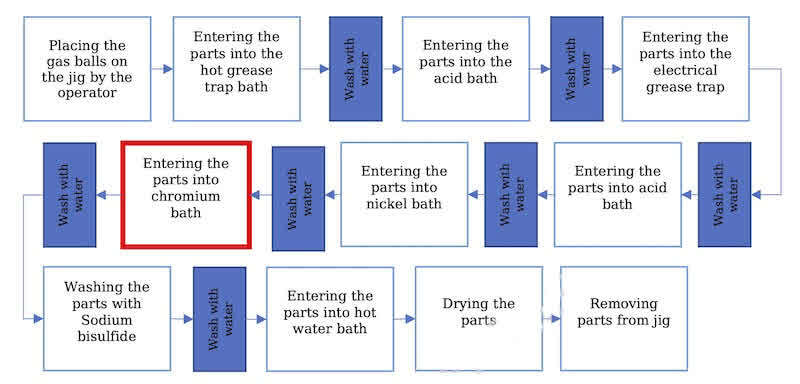
Fig. 1. The process of plating the balls in gas ball valves
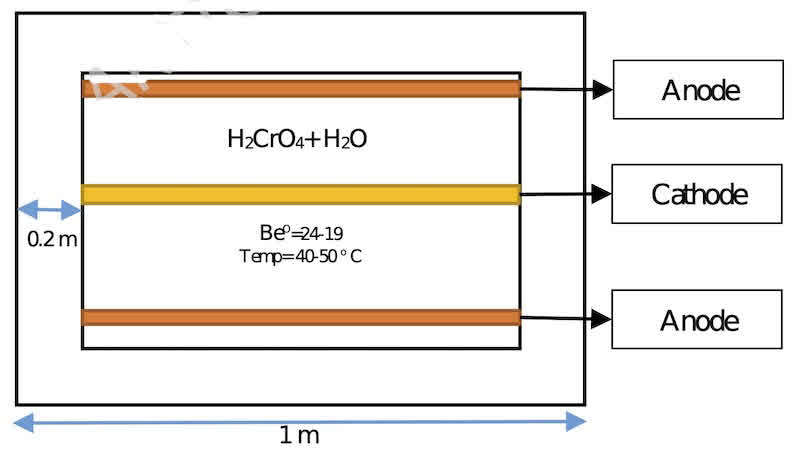
Fig. 2. Electroplating bath view from above
Materials
In this study, the plastic balls were used with two outer diameters of 4.9 cm (with an inner diameter of 3.8 cm) and 6.7 cm (with an inner diameter of 5.85 cm). Also, the parts to be electroplated are the balls used in the production of gas valves and they were used in two sizes of 0.5 inches and 1.5 inches (table 1 and 2).
Table 1. The plastic balls information
| Ball’s number | Material | Inner diameter | Outer diameter |
| Number 1 | Flexible plastic | 3.80 cm | 4.90 cm |
| Number 2 | Flexible plastic | 5.85 cm | 6.70 cm |
Table 2. The electroplated parts information
| Electroplated parts | Electroplating type | Dimensions |
| Balls in gas ball valves type 1 | chromium | 0.5 in |
| Balls in gas ball valves type 2 | chromium | 1.5 in |
Samples
In this study, 80 samples of air emitted from the surface of the electroplating bath were collected based on the NIOSH 7300 analytical method [13]. Collecting samples wTas based on table 3 and Figure 3. Sampling was done in two cases: 1. The surface of the tub without enclosure and with enclosure. The case where the four sides of the tub (north, south, east and west) are enclosed up to a height of 0.7 m (figure 4). Each cases has 8 different combination of other variable (number of plastic ball’s layer (figure 5), plastic ball’s number, ball in gas ball valve’s type) and 2 base modes which is according to not using plastic balls for electroplating two different types of balls in gas ball valves.
Table 3. Samples
| Enclosing 4 sides of bath | Without enclosing | |||
| Sampling mode | Balls in gas ball valves type 1 | Balls in gas ball valves type 2 | Balls in gas ball valves type 1 | Balls in gas ball valves type 2 |
| Base mode | 4 samples | 4 samples | 4 samples | 4 samples |
| Electroplating bath with 1 layer of plastic balls number 1 | 4 samples | 4 samples | 4 samples | 4 samples |
| Electroplating bath with 1 layer of plastic balls number 2 | 4 samples | 4 samples | 4 samples | 4 samples |
| Electroplating bath with 2 layer of plastic balls number 1 | 4 samples | 4 samples | 4 samples | 4 samples |
| Electroplating bath with 2 layer of plastic balls number 2 | 4 samples | 4 samples | 4 samples | 4 samples |
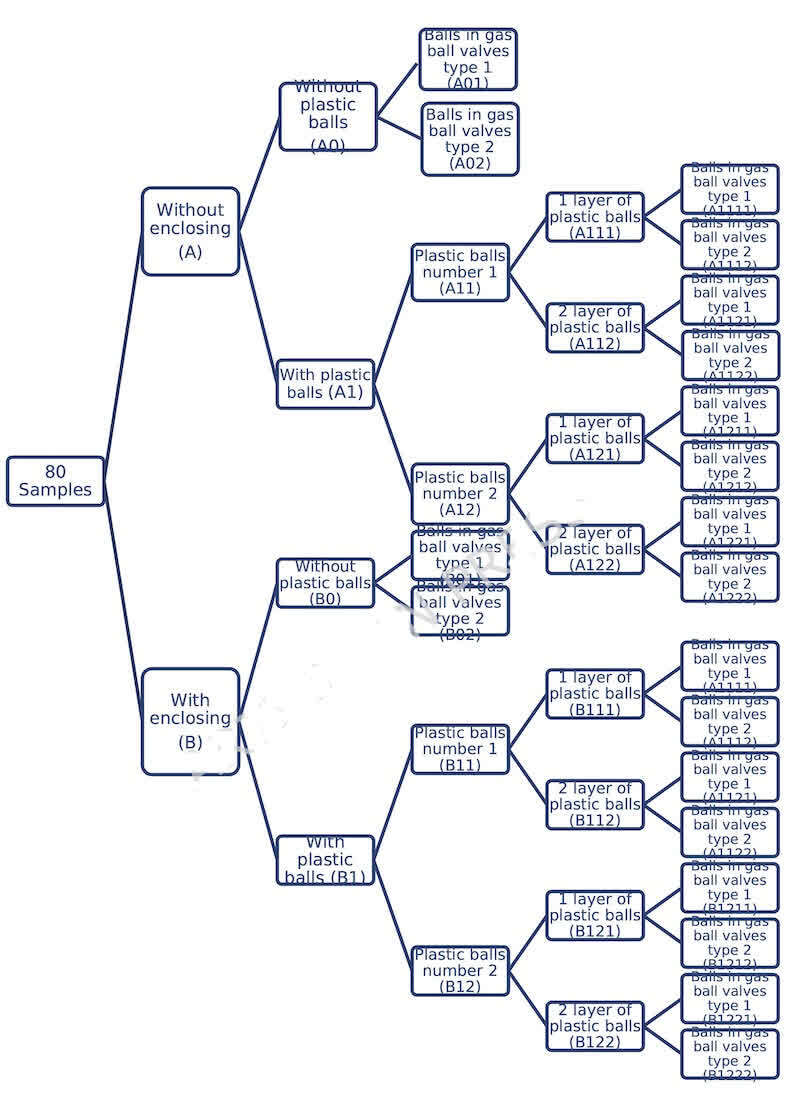
Fig. 3. The statistical population of the research
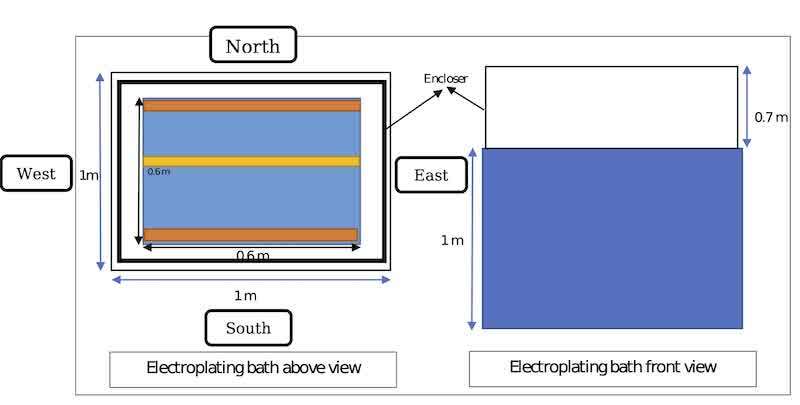
Fig. 4. Enclosing view of bath
Fig. 5. Two types of balls layer
Collecting samples way
In this sampling method and in accordance with the NIOSH 7300 analytical method, the sampling pump was first calibrated by a sampling filter in the laboratory using a rotameter and then set to a real flow rate of 3 liters per minute for 60 minutes. In the first step, in order to determine the concentration of the pollutant in the environment, 4 environmental samples were taken. In the second step, with other variables constant, two plastic balls type (number 1 and 2) were spread on the surface of the plating bath and the pollutant concentration was measured as in the first step. In the third step, with other variables being constant, the number of layers covered by balls was increased to two layers of balls and investigated. In the fourth step, with the number of layers covered by the balls and the type of balls being constant, the amount of pollutant concentration was investigated when two different types of balls in gas ball valves used for electroplating, and in the last step, by covering the four sides of the electroplating bath (north, south, east and west) up to a height of 0.7 m, all steps were repeated again. In all stages, the air flow speed at the distances, next to the bath, 0.5 m and 1 m from the plating bath measured using a thermal anemometer. the concentration of the solution inside the plating bath using a Baume meter, humidity, dry temperature and wet temperature using a thermal anemometer and the temperature of the plating bath was measured using an alcohol thermometer.
Measuring the amount of pollutants
After collecting the samples while ensuring compliance with the principles mentioned in the sampling method, they were analyzed to determine the amount of pollutant (chromium metal) using the VISTA-MPX model of the Inductively Coupled Plasma-Atomic Emission Spectrometer (ICP-AES).
Statistics
After collecting all the information, it was entered into SPSS Version 23 software and subjected to statistical analysis. In order to analyze the data, descriptive statistics methods such as mean, standard deviation and quartiles were used. Kolmogorov-Smirnov test was used to measure normality. T-test was used to measure the relationship between normal variables and Mann-Whitney test was used to measure non-normal variables. In addition, simple linear regression test was used.
Results and Discussion
In general, 80 pollutant samples released from the surface of the plating bath were collected according to NIOSH 7300 analytical method and included in this study, including 20 groups, each with 4 samples.
The average (standard deviation) concentration of the respiratory pollutant released from the electroplating bath is 29.59 (54.4) mg/m3 with a range of changes from 0.1 to 186.5 mg/m3. Descriptive results are shown in Table 4.
Table 4. Descriptive results
| Variable | Average | Standard deviation | Maximum | Minimum |
| Pollutant concentration released from electroplating bath (mg/m3) | 29.59 | 54.4 | 186.5 | <0.1 |
| Ambient humidity (percent) | 29.93 | 6.42 | 40 | 16.4 |
| Dry temperature (0C) | 20.29 | 1.55 | 23.8 | 17.9 |
| Wet temperature (0C) | 1.14 | 10.85 | 13.9 | 9.2 |
| Electroplating bath temperature (0C) | 43.3 | 2.11 | 47 | 40 |
| Concentration of the electroplating bath solution (g/l) | 19.5 | 2.51 | 22 | 17 |
The analytical results of the study show that the concentration of the pollutant released from the electroplating tub in the case of enclosure is significantly lower than the cases without enclosure (P<0.0001). In the same way, the results of the study indicate that the concentration of the pollutant released from the plating bath in the state where the surface of the bath is covered with plastic balls is significantly lower than the state without plastic balls (P=0.004). These results are similar to the results of Vasantha Kumar et al.'s study on the use of 4-inch black balls to cover the surface of the water tank [10]. Also, these results are consistent with Hassan and Pearson's studies on the use of these balls on the surface of Australian lakes [12].
This is despite the fact that the type of the balls in gas ball valves as well as the type of the plastic balls did not show a significant relationship with the concentration of pollutant emitted from the electroplating bath (Table 5).
The results of this study are similar to an invention in 1999 in the United States. In this invention, the surface of the electroplating bath is covered with a layer of foam (polymeric, metal or glass) and the amount of pollutant spread is measured. This invention estimates the performance of foams in the chrome plating industry up to 96 percent reduction of emitted pollutants. Also, this invention refers to research related to the use of balls on the surface of the bathtub and mentions the performance of these balls in reducing diffuse pollutants in the range between 50 and 60 percent. However, the balls used in the mentioned study had a specific diameter (30 mm) and the use of balls with different dimensions was not mentioned [14].
Table 5. Correlation of investigated factors with pollutant concentration
| Variable | Pollutant concentration (mg/m3) Average | Pollutant concentration (mg/m3) Average Standard Deviation | Comparison between two groups | |
| Enclosing | With encloser | 3.04 | 3.62 | P < 0.0001 |
| Enclosing | Without encloser | 55.46 | 67.18 | P < 0.0001 |
| Plastic balls | With balls | 26.67 | 53.07 | P = 0.004 |
| Plastic balls | Without balls | 41.66 | 59.99 | P = 0.004 |
| Balls in gas ball valves | Type 1 | 36.42 | 62.87 | P = 0.743 |
| Balls in gas ball valves | Type 2 | 22.59 | 43.8 | P = 0.743 |
| Number of ball layers | One | 30.61 | 56.49 | P = 0.074 |
| Number of ball layers | Two | 22.48 | 49.76 | P = 0.074 |
| Number of plastic balls | One | 26.61 | 53.3 | P = 0.353 |
| Number of plastic balls | Two | 26.73 | 53.71 | P = 0.353 |
In this study, the effect of air flow speed around the electroplating bath was evaluated and its results are shown in Table 6. Based on the results of this table, the increase in air speed near the bath from the north, east, and west significantly increases the concentration of pollutants emitted from the electroplating bath. Meanwhile, the air speed near the bath from the south increases the concentration of the pollutant emitted from the electroplating bath, but this amount is not statistically significant.
In addition, the results of this study showed that the air velocity at a distance of 0.5 m and 1 m does not have a significant effect on the amount of pollutant concentration emitted from the electroplating bath. Air speed at a distance of 0.5 m, north and east leads to an increase in pollutant concentration and air speed at a distance of 0.5 m, west and south causes a decrease in pollutant concentration. But none of them are meaningful.
Also, the air speed at a distance of 1 m, north and west, leads to an increase in the concentration of pollutants, and also the air speed at a distance of 1 m, east and south, causes a decrease in the concentration of pollutants, and these results are not statistically significant (Table 6).
Table 6. The effect of the air flow speed around the electroplating bath on the pollutant concentration emitted from the electroplating bath
| Variable | B2 | Standard Error | Beta standard coefficient1 | t | sig |
| Air velocity near the bath; north | 0.505 | 0.195 | 0.286 | 2.586 | 0.012 |
| Air velocity near the bath; east | 3.647 | 1.432 | 0.282 | 2.546 | 0.013 |
| Air velocity near the bath; west | 3.923 | 1.646 | 0.265 | 2.383 | 0.020 |
| Air velocity near the bath; south | 1.281 | 1.439 | 0.102 | 0.89 | 0.376 |
| Air velocity at a distance of 0.5 m; north | 0.676 | 0.533 | 0.145 | 1.270 | 0.208 |
| Air velocity at a distance of 0.5 m; east | 2.163 | 1.513 | 0.163 | 1.430 | 0.157 |
| Air velocity at a distance of 0.5 m; west | -2.760 | 2.042 | -0.154 | -1.351 | 0.181 |
| Air velocity at a distance of 0.5 m; south | -0.634 | 0.590 | -0.123 | -1.074 | 0.286 |
| Air velocity at a distance of 1 m; north | 6.006 | 12.012 | 0.082 | 0.50 | 0.620 |
| Air velocity at a distance of 1 m; east | -0.507 | 1.424 | -0.058 | -0.356 | 0.724 |
| Air velocity at a distance of 1 m; west | 9.651 | 13.685 | 0.115 | 0.705 | 0.485 |
| Air velocity at a distance of 1 m; south | -2.17 | 3.530 | -0.101 | -0.615 | 0.542 |
1 Beta is a standardized coefficient between -1 to +1 in range and show the strength of the prediction; 2 B is an unstandardized coefficient which means original units besides the slope and tell if the independent variable is a significant predictor of the dependent variable.
The results from Table 7 show that with the increase in the bath temperature, the concentration of the pollutant emitted from the plating bath increases significantly (P<0.0001). In addition, the increase in the concentration of the bath solution is significantly related to the decrease in the concentration of the pollutant released from the plating bath (P<0.0001).
The results of this study showed that increasing the temperature of the electroplating bath is one of the most effective factors in the amount of pollutants emitted from the electroplating bath. Considering the point that the primary function of these balls is to prevent temperature loss in electroplating bath [4], these results confirm the performance of balls in reducing the amount of pollutants released from the surface of the electroplating bath.
Meanwhile, the results show that an increase in ambient humidity, an increase in wet temperature, and an increase in dry temperature all lead to an increase in the concentration of pollutants emitted from the plating bath. But this increase is slight and none of them are statistically significant.
Table 7. The effect of different factors on the concentration of the pollutant emitted from the electroplating bath
| Variable | B | Standard Error | Beta standard coefficient | t | sig |
| Ambient humidity | 0.513 | 1.009 | 0.059 | 0.509 | 0.612 |
| Dry temperature | 1.669 | 3.993 | 0.048 | 0.418 | 0.677 |
| Wet temperature | 3.438 | 5.681 | 0.070 | 0.605 | 0.547 |
| Electroplating bath temperature | 10.025 | 2.736 | 0.390 | 3.664 | <0.0001 |
| Concentration of the electroplating bath solution | - 10.483 | 2.183 | -0.485 | - 4.801 | <0.0001 |
Limitation
This study has several limitations. As a limitation, this research were carried out in a single chrome-plating facility, which may restrict the applicability of these findings to other plating operations that utilize different ventilation configurations, tank designs, or operational procedures. Moreover, while various parameters were assessed, certain potentially significant factors—such as air turbulence above the plating bath, fluctuations in the performance of local exhaust ventilation, and the degree of mixing within the bath—were not directly measured and may have impacted vapor emission levels.
Conclusions
According to the findings of this research, it can be stated that the plastic balls covering the surface of the electroplating bath are an effective way to reduce the amount of pollutants emitted from the surface of the electroplating bath. The size of this balls and the number of layers covered by them did not have a significant difference in the amount of pollutants emitted from the surface of the electroplating bath but it might have indirect impact by increasing the bath’s temperature which needs more research, so therefore it can be concluded that the important parameters in choosing them are not getting stuck between the electroplating jigs and not causing disruption to the speed of the process. And also it could be concluded that enclosure around the electroplating, by limiting the speed of air flow around the plating tank can be an effective factor in reducing the amount of pollutants spread from the surface of the tank. Therefore, decreased air velocity (< 0.15 m/s) can reduce the emission of pollutants from the surface of the tank.
Written by Parnian Ilbag1, Masoud Rismanchian2, and Saeid Yazdanirad3
- Department of Occupational Health Engineering, School of Public Health and Nutrition Isfahan University of Medical Sciences, Isfahan, Iran. Email: parnian.rcsjp@gmail.com. ORCID: 0000-0003-4300-133X.
- Department of Occupational Health Engineering, School of Public Health and Nutrition Isfahan University of Medical Sciences, Isfahan, Iran. Email: rismanchian@hlth.mui.ac.ir. ORCID: 0000-0001-8843-9445.
- Department of Occupational Health, School of Health, Shahrekord University of Medical Sciences, Isfahan, Iran. Email: saeedyazdanirad@gmail.com. ORCID: 0000-0002-5251-6637.
Declarations
Ethics approval and consent to participate: This study was approved as a research project by the ethics committee of Isfahan University of Medical Sciences (IR.MUI.RESEARCH.REC.1399.558). All methods were performed in accordance with relevant guidelines and regulations. All subjects gave their written informed consent to participate.
Consent for publication: Not applicable.
Competing interests: The authors declare that they have no competing interests.
Funding: This study was supported by Isfahan University of Medical Sciences.
Authors’ contributions: Parnian Ilbag: Data Collection, Methodology, Formal analysis, Investigation, Writing – original draft, and Visualization. Masoud Rismanchian: Investigation, Methodology, Writing – original draft, Writing – review & editing. Saeid Yazdanirad: Writing – original draft, Writing – review & editing, and Visualization.
Acknowledgements: Special thanks to Isfahan University of Medical Sciences for supporting this document.
Availability of data and materials: The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
References
- Alamri AH. Localized corrosion and mitigation approach of steel materials used in oil and gas pipelines–An overview. Engineering failure analysis. 2020;116:104735.
- Kania H. Corrosion and anticorrosion of alloys/metals: the important global issue. MDPI; 2023. p. 216.
- Hamza NA, Majeed AS, Jawd SM. Review on types and methods of electroplating on metals. J Power Electron Devices. 2021;7:44-51.
- Association SE. Hexavalent chromium in electroplating: Prevention and Control of Chromic Acid Mist 2018.
- Khoshakhlagh AH, Ghobakhloo S, Peijnenburg WJ, Gruszecka-Kosowska A, Cicchella D. To breathe or not to breathe: Inhalational exposure to heavy metals and related health risk. Science of the Total Environment. 2024;932:172556.
- Khoshakhlagh AH, Mohammadzadeh M, Bamel U, Gruszecka-Kosowska A. Human exposure to heavy metals and related cancer development: a bibliometric analysis. Environmental Science and Pollution Research. 2023;30(51):109867-88.
- Kirichenko KY, Vakhniuk I, Ivanov V, Tarasenko I, Kosyanov DY, Medvedev S, et al. Complex study of air pollution in electroplating workshop. Scientific reports. 2020;10(1):11282.
- Saunders J, Coldwell M, Bennett S, editors. Comparison of local exhaust ventilation with other exposure controls used in electrolytic nickel plating. E3S Web of Conferences; 2025: EDP Sciences.
- Wang LK, Wang M-HS. Environmental management of electroplating and metal-finishing operations. Control of heavy metals in the environment: CRC Press; 2025. p. 325-78.
- VASANTHA KUMAR P. Reduction of Water Vapour by Using Shade Balls. International Journal of Engineering and Techniques. 2018;4(2):2-5.
- Waheeb Youssef Y, Khodzinskaya A. A Review of Evaporation Reduction Methods from Water Surfaces. E3S Web Conf. 2019;97:05044.
- Hassan MM, Peirson WL. Evaporation mitigation by floating modular devices. IOP Conference Series: Earth and Environmental Science. 2016;35:012022. 10.1088/1755-1315/35/1/012022.
- Kim D, Jung S, Yoon C. Evaluation of airborne total suspended particulates and heavy metals in anodizing and electroplating surface treatment process. Scientific Reports. 2021;11(1):22537.
- Bourke SH. Control agent for reducing metal acid mist emissions from electrolytic cell operations. Google Patents; 1999.




































